เมื่อมีเวลาเล่น sagame168th ไม่กี่นาที คนเล่นจะตกลงใจเล่นอย่างไร?
 การเล่นบาคาร่ากับ sagame168 ในตอนนี้นั้นแตกต่างจากเมื่อก่อนมากเลยนะครับ ครั้งก่อนเราอาจจะเป็นไปได้ว่าจะมีเวลาเล่นหลายชั่วโมง แม้กระนั้นในขณะนี้เพียงแค่หาเวลาว่างเปิดเกมก็ยากแล้ว เพราะฉะนั้น หลายท่านจึงเลือกเล่นบาคาร่าระหว่างพักตอนกลางวัน ระหว่างรอคอยประชุม ระหว่างเดินทาง หรือในขณะสั้นๆก่อนที่จะไปทำกิจกรรมอื่นต่อกันเพิ่มขึ้นเรื่อยๆ เมื่อเวลามีจำกัดแบบงี้ มันทำให้พวกเรามีแนวทางคิดหรือตัดสินใจแตกต่างจากตอนที่มีเวลาเล่นนานๆหรือไม่? แล้วพวกเราจะตกลงใจเล่นยังไงดี? ในบทความนี้ เราจะมาดูกันเลยครับผม!
การเล่นบาคาร่ากับ sagame168 ในตอนนี้นั้นแตกต่างจากเมื่อก่อนมากเลยนะครับ ครั้งก่อนเราอาจจะเป็นไปได้ว่าจะมีเวลาเล่นหลายชั่วโมง แม้กระนั้นในขณะนี้เพียงแค่หาเวลาว่างเปิดเกมก็ยากแล้ว เพราะฉะนั้น หลายท่านจึงเลือกเล่นบาคาร่าระหว่างพักตอนกลางวัน ระหว่างรอคอยประชุม ระหว่างเดินทาง หรือในขณะสั้นๆก่อนที่จะไปทำกิจกรรมอื่นต่อกันเพิ่มขึ้นเรื่อยๆ เมื่อเวลามีจำกัดแบบงี้ มันทำให้พวกเรามีแนวทางคิดหรือตัดสินใจแตกต่างจากตอนที่มีเวลาเล่นนานๆหรือไม่? แล้วพวกเราจะตกลงใจเล่นยังไงดี? ในบทความนี้ เราจะมาดูกันเลยครับผม!
 เวลาจำกัดสำหรับในการเล่น sagame168 ทำให้โฟกัสกับสิ่งสำคัญเยอะขึ้น
เวลาจำกัดสำหรับในการเล่น sagame168 ทำให้โฟกัสกับสิ่งสำคัญเยอะขึ้น
เมื่อเรามีเวลาจำกัดสำหรับในการเล่น sagame168th พวกเราก็จำต้องยอมลดรายละเอียดอะไรบางอย่างลงโดยทันที รวมทั้งหันไปให้ความสำคัญกับสิ่งที่สำคัญและก็จำเป็นจะต้องจริงๆเพียงแค่นั้น ซึ่งมันมิได้มีเหตุมาจากความไม่รอบคอบเลยนะครับผม แต่มันเป็นการจัดลำดับที่ดีกว่านั่นเอง
• เลือกมองข้อมูลที่เคยชินก่อน: ถ้าเกิดมีเวลาไม่กี่นาที ผมเสนอแนะให้ทดลองดูหรือประเมินเหตุการณ์จากสิ่งที่คุ้นเคยก่อนเสมอ ไม่ว่าจะเป็นรูปแบบการดูเกม การสังเกตจังหวะ หรือหนทางที่เคยใช้มาก่อน จะทำให้ตกลงใจได้เร็วขึ้น
• ลดขั้นขณะที่ไม่สำคัญ: ในเมื่อมีเวลาจำกัด เราคงไม่ต้องการที่จะอยากเสียเวล่ำเวลากับรายละเอียดเยอะมากอยู่แล้ว เราก็แค่ตัดเรื่องที่ไม่เกี่ยวกับเป้าหมายออกไปก่อน เพื่อจุดโฟกัสกับสิ่งที่สำคัญที่สุดได้จริงๆมันจะช่วยทำให้ตัดสินใจได้อย่างเที่ยงตรงขึ้น
• ให้ความสำคัญกับความแน่ชัด: ในระยะเวลาสั้นๆเราจะรู้สึกสุขใจกับข้อมูลที่เข้าใจง่ายมากกว่าข้อมูลที่จำเป็นต้องคิดหรือพินิจพิจารณาหลายอย่างพร้อมกัน ยิ่งมีเวลาน้อยก็ยิ่งต้องตกลงใจจากสิ่งที่แลเห็นได้โดยทันทีมากกว่าเท่านั้นเลยครับผม
• ดูเป้าหมายระยะสั้น: ในเมื่อพวกเรามีเวลาเล่น sagame สั้นๆพวกเราก็เปลี่ยนแปลงจากมองภาพรวมระยะยาวมาจุดโฟกัสวัตถุประสงค์ระยะสั้นในตอนนั้นก็พอแล้วนะครับ มันจะทำให้ข้อจำกัดด้านเวลาเปลี่ยนเป็นกรอบการเล่นที่ดีไปเลยล่ะครับ
ขณะพักสั้นๆเปลี่ยนรูปแบบการตัดสินใจเล่น sagame มากกว่าที่คิด
คนจำนวนไม่น้อยอาจมีความคิดว่า หากมีเวลาเล่น sagame แค่ช่วงสั้นๆนั้นจะมีผลให้เล่นเร็วขึ้น แม้กระนั้นอันที่จริงแล้ว วิธีคิด การตัดสินใจ หรือการคาดการณ์เหตุการณ์ของพวกเราจะเปลี่ยนไปด้วย ทำให้การตัดสินใจมีลักษณะเฉพาะในระยะเวลาจำกัดอย่างนี้เลยครับ
• ความเกี่ยวเนื่องคือปัจจัยสำคัญ: เพียงพอพวกเรามีเวลาเล่นไม่นาน พวกเราจะไม่ต้องการที่จะอยากเสียเวลาเริ่มใหม่ พวกเราจึงให้ความเอาใจใส่กับความเกี่ยวเนื่องของการเล่นมากยิ่งกว่าแปลงทางไปเรื่อยๆส่วนมากก็เลยยึดวิถีทางสำหรับการเล่นแบบเดียวไปเลย
• ความไม่แน่ใจลดลง: เวลาเป็นข้อกำหนดที่สำคัญมาก ผู้คนจำนวนมากพบว่าใช้เวลาตกลงใจลดน้อยลงกว่าธรรมดาด้วย ซึ่งมันไม่ใช่ว่ามั่นใจมากจนเกินความจำเป็นครับ แต่เพราะเหตุว่าไม่ว่างกลับไปคิดหรือลังเลอีก มันก็เลยทำให้ดูอย่างกับว่าพวกเราตัดสินใจเร็วขึ้นนั่นเอง
• ไม่ได้โฟกัสแต่เกม: การเล่นตอนพักหรือขณะที่ทำกิจกรรมอื่นๆมันทำให้ความพอใจของพวกเราสำหรับการเล่น sagame168 มิได้อยู่ที่บาคาร่าสิ่งเดียวด้วย เพียงพอต้องตัดสินใจในสภาพแวดล้อมที่มีสิ่งรบกวนมาก มันก็มีผลต่อวิธีคิดด้วย
• ความสบายมีผลต่อการเลือก: เมื่อมีเวลาน้อย พวกเรามักจะเลือกแนวทางที่ทำให้พวกเราตัดสินใจได้ง่ายที่สุด ซึ่งมันเป็นเรื่องปกติของมนุษย์เลยล่ะครับ โดยยิ่งไปกว่านั้นเมื่อสมองของพวกเราอุตสาหะลดภาระหน้าที่การคิดอะไรก็ตามภายใต้ข้อจำกัดของเวลา
คนเล่น sagame 168th ในปัจจุบันปรับพฤติกรรมตามเวลาที่มีเพิ่มขึ้นเรื่อยๆ
แน่นอนว่า คนเล่น sagame168 มากมายมิได้เห็นว่าขณะที่จำกัดเป็นปัญหาเสมอไปนะครับ เพราะเหตุว่าหลายท่านสามารถปรับพฤติกรรมแล้วก็ปรับปรุงวิธีเล่นหรือแนวทางคิดให้เข้ากับแบบการใช้ชีวิตของตนได้ด้วย ทำให้เล่นได้ดิบได้ดีขึ้นในเวลาจำกัดไปเลย
• กำหนดขอบเขตการเล่น: เมื่อเราเล่นในช่วงพักเป็นประจำเราจะเริ่มรู้จักกับการกำหนดขอบเขตของตัวเองไปเลย ไม่ว่าจะเป็นเวลาที่ใช้หรือวัตถุประสงค์ในการเล่น ทำให้เราจัดสรรเวลาให้กับกิจกรรมในแต่ละวันเจริญมากยิ่งขึ้นเลยนะครับ
• วิวัฒนาการตัดสินใจภายใต้ข้อกำหนด: การจำต้องเลือกภายในช่วงเวลาที่มีอยู่น้อยนิด มันก็ทำให้หลายๆคนทำความเข้าใจและพัฒนา
การตัดสินใจในเวลาอย่างงี้ก้าวหน้ามาก เพียงพอพวกเราเจอสถานการณ์นี้บ่อยๆเราก็จะยิ่งจัดการกับมันได้ดีขึ้นด้วยครับ
• ปรับวัตถุประสงค์ให้เข้ากับสถานการณ์จริง: sagame168 ผู้ที่เล่นSagame168th ช่วงสั้นๆจะเข้าใจเลยว่า เป้าหมายที่ตั้งเอาไว้ควรต้องสอดคล้องกับในเวลาที่มีด้วย หากพวกเรากำหนดเป้าหมายให้เหมาะสมก็จะทำให้การตัดสินใจดียิ่งขึ้น แล้วก็ยังลดแรงกดดันที่บางทีอาจเกิดขึ้นได้อีกด้วย
เมื่อมีเวลาไม่กี่นาที การตัดสินใจเล่น sagame 168 ก็มีคุณภาพมากขึ้นได้
ในตอนที่มีจำกัดมิได้ทำให้พวกเราเล่น sagame168 มิได้หรือแย่ลงครับผม แต่มันทำให้เราสามารถทำความเข้าใจการจัดลำดับความสำคัญ โฟกัสกับสิ่งที่สำคัญ และก็ปรับแนวทางคิดให้กับสถานการณ์จริงๆได้มากขึ้นด้วย เราไม่มีความจำเป็นที่ต้องเล่นให้เป๊ะราวกับที่เคยทำมาตลอด ไม่จำเป็นต้องทำทุกอย่างพร้อมเพียงกัน เพียงแค่เลือกใช้เวลาไปกับแผนการที่มี รู้เรื่องการกระทำและก็สไตล์ของตัวเองให้มากขึ้น พวกเราก็จะมองเห็นได้เลยว่า เวลาไม่ใช่ข้อกำหนดในการเล่นsagame168th ถ้าพวกเราใช้เวลาที่มีให้กำเนิดผลดีสูงที่สุดได้
บาคาร่าเว็บใหญ่ sagame168 sagame168th1.co 30 สิงหาคม 2569 Juliane casino sagame แตกหนัก Top 67
ขอขอบคุณreference sagame168
https://rebrand.ly/sagame168th-1





 ความรู้สึกว่า เกม live22slot เกมนี้คุ้นมือ แม้ว่าจะยังไม่เคยเล่นมาก่อนก็ตาม เป็นผลมาจากอะไร?
ความรู้สึกว่า เกม live22slot เกมนี้คุ้นมือ แม้ว่าจะยังไม่เคยเล่นมาก่อนก็ตาม เป็นผลมาจากอะไร? เพราะอะไรบางเกมของ live22 ถึงทำให้พวกเราปรับตัวแล้วก็คุ้นมือได้เร็ว แม้จะเล่นครั้งแรก?
เพราะอะไรบางเกมของ live22 ถึงทำให้พวกเราปรับตัวแล้วก็คุ้นมือได้เร็ว แม้จะเล่นครั้งแรก?
 • Joker Gaming: ค่ายเก๋าเกมระดับตำนาน หากแม้ภาพบางทีอาจจะไม่หวือหวาเท่าค่ายใหม่ๆแต่ว่าเรื่องความคลาสสิกและโบนัสที่แตกแบบเนื้อๆเน้นย้ำๆยังคงเชื่อใจได้เสมอ
• Joker Gaming: ค่ายเก๋าเกมระดับตำนาน หากแม้ภาพบางทีอาจจะไม่หวือหวาเท่าค่ายใหม่ๆแต่ว่าเรื่องความคลาสสิกและโบนัสที่แตกแบบเนื้อๆเน้นย้ำๆยังคงเชื่อใจได้เสมอ มันไม่ใช่แค่เรื่องของเงินรางวัลที่แตกง่ายอย่างเดียว แม้กระนั้นมันเป็นความสุขใจที่คุณจะได้รับ ระบบฝาก-ถอนออโต้ที่รวดเร็วด้านในไม่กี่วินาที ความยั่งยืนทางการเงินที่คุณเชื่อมั่นได้ว่าเล่นได้หลักแสนหลักล้านก็ถอนได้จริง ตลอดจนความมากมายหลายของเกมที่มีให้เลือกเล่นแบบไม่สิ้นสุด ทั้งยังสล็อต บาคาร่า รวมทั้งคาสิโนสดอื่นๆ
มันไม่ใช่แค่เรื่องของเงินรางวัลที่แตกง่ายอย่างเดียว แม้กระนั้นมันเป็นความสุขใจที่คุณจะได้รับ ระบบฝาก-ถอนออโต้ที่รวดเร็วด้านในไม่กี่วินาที ความยั่งยืนทางการเงินที่คุณเชื่อมั่นได้ว่าเล่นได้หลักแสนหลักล้านก็ถอนได้จริง ตลอดจนความมากมายหลายของเกมที่มีให้เลือกเล่นแบบไม่สิ้นสุด ทั้งยังสล็อต บาคาร่า รวมทั้งคาสิโนสดอื่นๆ สุดท้ายนี้ การพนันมีการเสี่ยง สล็อต168 ผู้เล่นควรจะเล่นอย่างมีสติสัมปชัญญะแล้วก็ใช้เงินเย็นสำหรับเพื่อการเล่นเสมอครับผม ถ้าเกิดคุณกำลังมองหาเว็บคู่ใจดีๆสักเว็บ ลองเปิดใจให้เว็บแห่งนี้มองสักครั้ง แล้วคุณจะเข้าหัวใจว่าเว็บไซต์ตรงของจริงเขาเล่นกันอย่างไรครับผม!
สุดท้ายนี้ การพนันมีการเสี่ยง สล็อต168 ผู้เล่นควรจะเล่นอย่างมีสติสัมปชัญญะแล้วก็ใช้เงินเย็นสำหรับเพื่อการเล่นเสมอครับผม ถ้าเกิดคุณกำลังมองหาเว็บคู่ใจดีๆสักเว็บ ลองเปิดใจให้เว็บแห่งนี้มองสักครั้ง แล้วคุณจะเข้าหัวใจว่าเว็บไซต์ตรงของจริงเขาเล่นกันอย่างไรครับผม!
 สวัสดีขอรับเพื่อนพ้องๆพี่ๆน้องๆสายปั่นทุกคน วันนี้ผมต้องการจะมาแชร์ประสบการณ์ตรงจากผู้ที่เป็น “สายปั่นตัวจริง” ที่อยู่ในวงการเกมพนันออนไลน์มาอย่างยาวนาน เจ็บมามากมาย เจอกลลวงมาก็แยะ แต่ว่าล่าสุดผมได้ไปพบกับพิกัดลับ (ที่ไม่ลับอีกต่อไป) อย่างเว็บไซต์ สล็อตเว็บตรง โบนัสแตกกระจาย ทดสอบเล่นฟรี ถอนได้
สวัสดีขอรับเพื่อนพ้องๆพี่ๆน้องๆสายปั่นทุกคน วันนี้ผมต้องการจะมาแชร์ประสบการณ์ตรงจากผู้ที่เป็น “สายปั่นตัวจริง” ที่อยู่ในวงการเกมพนันออนไลน์มาอย่างยาวนาน เจ็บมามากมาย เจอกลลวงมาก็แยะ แต่ว่าล่าสุดผมได้ไปพบกับพิกัดลับ (ที่ไม่ลับอีกต่อไป) อย่างเว็บไซต์ สล็อตเว็บตรง โบนัสแตกกระจาย ทดสอบเล่นฟรี ถอนได้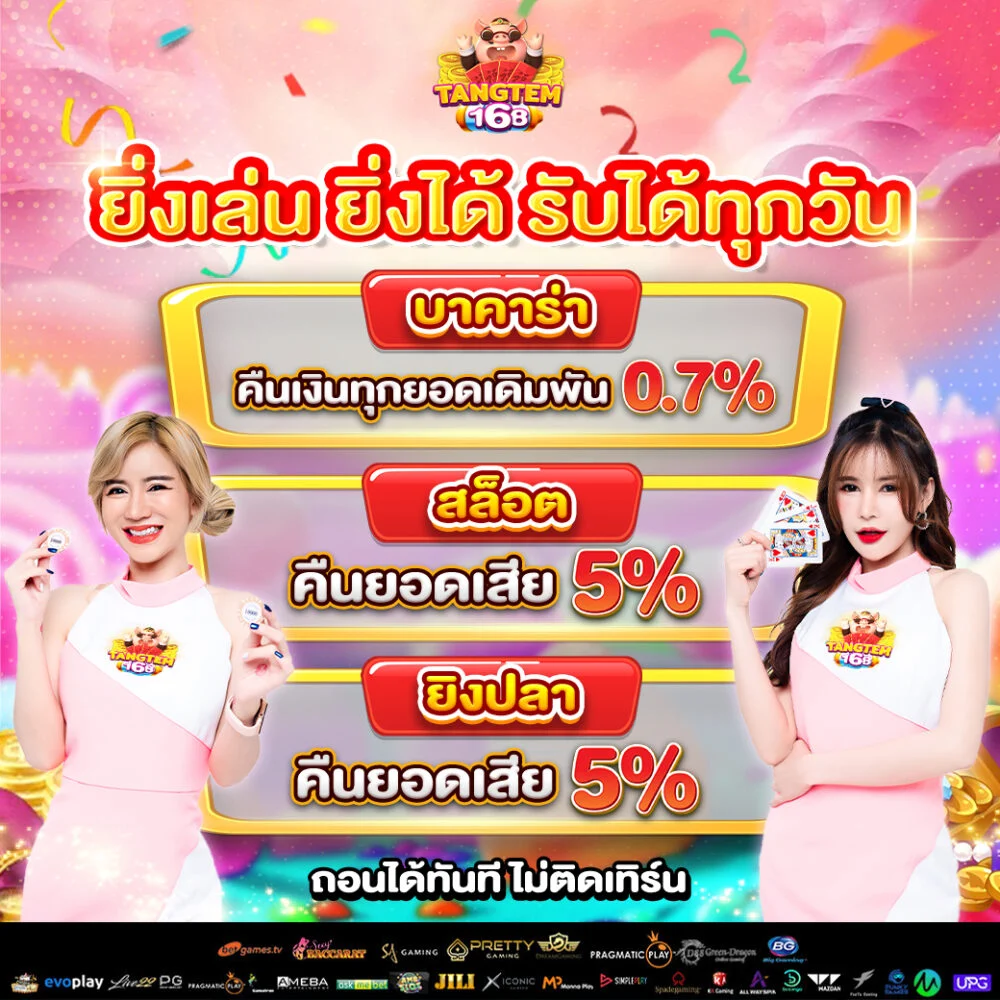 ซึ่งบอกได้คำเดียวเลยว่าทำเอาคนเล่นสล็อตรวมทั้งเกมพนันอย่างผมถึงกับต้องอุทานว่า “ไปอยู่ไหนมาทำไมเพิ่งพบ!” เนื้อหานี้ผมจะมารีทิวทัศน์แบบเจาะลึกจากสิ่งที่ผมพบมากับตัว ไม่มีการอวยไส้แตก แม้กระนั้นจะเล่าให้ฟังชัดๆว่าเพราะเหตุใดเว็บไซต์นี้ถึงเปลี่ยนเป็นกระแสในหมู่สายปั่นขณะนี้นะครับ
ซึ่งบอกได้คำเดียวเลยว่าทำเอาคนเล่นสล็อตรวมทั้งเกมพนันอย่างผมถึงกับต้องอุทานว่า “ไปอยู่ไหนมาทำไมเพิ่งพบ!” เนื้อหานี้ผมจะมารีทิวทัศน์แบบเจาะลึกจากสิ่งที่ผมพบมากับตัว ไม่มีการอวยไส้แตก แม้กระนั้นจะเล่าให้ฟังชัดๆว่าเพราะเหตุใดเว็บไซต์นี้ถึงเปลี่ยนเป็นกระแสในหมู่สายปั่นขณะนี้นะครับ เว็บไซต์บาคาร่า sexy168 เว็บไซต์ตรง ภาพคมชัด 4K มองกล้องสดแบบสับ ไม่มีประวัติความเป็นมาทุจริต !
เว็บไซต์บาคาร่า sexy168 เว็บไซต์ตรง ภาพคมชัด 4K มองกล้องสดแบบสับ ไม่มีประวัติความเป็นมาทุจริต ! ยอมรับมาซะดีๆว่าเวลาเราก้าวเท้าไปสู่วงการพนันออนไลน์ สิ่งที่ทำให้เราเสียอารมณ์ที่สุดคืออะไร? ไม่ใช่เล่นเสียหรอกจ้ะ ด้วยเหตุว่าประเด็นนั้นเราบริหารทุนได้ แต่ว่าคือน่ารำคาญหัวใจกับระบบเว็บไซต์ที่กระตุก ถ่ายทอดสดฟีลลิ่งเหมือนใช้กล้องถ่ายรูปโทรศัพท์มือถือปุ่มกดถ่าย ภาพแตกเป็นพิกเซลกระทั่งดูแต้มไพ่ไม่รู้เรื่อง แถมดีลเลอร์สาวสวยสับไพ่ทีหน้าทีหลังเบลอไปครึ่งหน้าจอ! หยุดทนกับประสบการณ์ห่วยๆอย่างงั้นได้แล้วค่ะ เพราะว่าวันนี้ตัวแม่จะพาวาร์ปมาทำความรู้จักกับที่สุดของความพรีเมียม นั่นก็คือ เว็บบาคาร่า168
ยอมรับมาซะดีๆว่าเวลาเราก้าวเท้าไปสู่วงการพนันออนไลน์ สิ่งที่ทำให้เราเสียอารมณ์ที่สุดคืออะไร? ไม่ใช่เล่นเสียหรอกจ้ะ ด้วยเหตุว่าประเด็นนั้นเราบริหารทุนได้ แต่ว่าคือน่ารำคาญหัวใจกับระบบเว็บไซต์ที่กระตุก ถ่ายทอดสดฟีลลิ่งเหมือนใช้กล้องถ่ายรูปโทรศัพท์มือถือปุ่มกดถ่าย ภาพแตกเป็นพิกเซลกระทั่งดูแต้มไพ่ไม่รู้เรื่อง แถมดีลเลอร์สาวสวยสับไพ่ทีหน้าทีหลังเบลอไปครึ่งหน้าจอ! หยุดทนกับประสบการณ์ห่วยๆอย่างงั้นได้แล้วค่ะ เพราะว่าวันนี้ตัวแม่จะพาวาร์ปมาทำความรู้จักกับที่สุดของความพรีเมียม นั่นก็คือ เว็บบาคาร่า168 บอกเลยว่าวินาทีนี้ ถ้าหากคนไหนกันยังไม่รู้จักชื่อนี้ นับว่าคุณพลาดความสนุกสนานระดับ High-End ไปอย่างโชคร้าย เพราะเหตุว่านี่คือ เว็บตรง ไม่ผ่านเอเย่นต์ที่ทุบทุกสถิติระบบหลังบ้าน ยกระดับเกมไพ่บาคาร่าให้กลายเป็นโรงภาพยนตร์ส่วนตัวด้วยสัญญาณสตรีมมิ่งสดส่งตรงจากคาสิโนต่างถิ่นด้วย ภาพชัดเจนระดับ 4K แจ้งชัดระดับที่เห็นดีเทลชุดบิกินี่ของดีลเลอร์ ชัดจนเห็นหน้าไพ่แบบไม่ต้องจ้องดูให้รอยเท้ากาขึ้น!
บอกเลยว่าวินาทีนี้ ถ้าหากคนไหนกันยังไม่รู้จักชื่อนี้ นับว่าคุณพลาดความสนุกสนานระดับ High-End ไปอย่างโชคร้าย เพราะเหตุว่านี่คือ เว็บตรง ไม่ผ่านเอเย่นต์ที่ทุบทุกสถิติระบบหลังบ้าน ยกระดับเกมไพ่บาคาร่าให้กลายเป็นโรงภาพยนตร์ส่วนตัวด้วยสัญญาณสตรีมมิ่งสดส่งตรงจากคาสิโนต่างถิ่นด้วย ภาพชัดเจนระดับ 4K แจ้งชัดระดับที่เห็นดีเทลชุดบิกินี่ของดีลเลอร์ ชัดจนเห็นหน้าไพ่แบบไม่ต้องจ้องดูให้รอยเท้ากาขึ้น! ในฐานะที่ผมเป็น “คนใน” ที่อยู่ในวงการเกมสล็อตออนไลน์มานานกว่า 5 ปี ลองถูกลองผิดมานับไม่ถ้วน เจ็บมาเยอะ และปังมาก็แยะ วันนี้ผมต้องการจะมาถ่ายทอดประสบการณ์ตรงแบบไม่มีกั๊ก ว่าเพราะเหตุใดค่ายเกมสุดคลาสสิกอย่าง สล็อตxo ถึงยังคงยืนหนึ่งในใจนักพนันคนประเทศไทย และมันคุ้มจริงไหมที่คุณจะเอาเงินและก็เวลามาลงทุนกับที่นี่ครับ
ในฐานะที่ผมเป็น “คนใน” ที่อยู่ในวงการเกมสล็อตออนไลน์มานานกว่า 5 ปี ลองถูกลองผิดมานับไม่ถ้วน เจ็บมาเยอะ และปังมาก็แยะ วันนี้ผมต้องการจะมาถ่ายทอดประสบการณ์ตรงแบบไม่มีกั๊ก ว่าเพราะเหตุใดค่ายเกมสุดคลาสสิกอย่าง สล็อตxo ถึงยังคงยืนหนึ่งในใจนักพนันคนประเทศไทย และมันคุ้มจริงไหมที่คุณจะเอาเงินและก็เวลามาลงทุนกับที่นี่ครับ • Black Beard Legacy:
• Black Beard Legacy:  slotxo การแจกเครดิตฟรีไม่ใช่การหลอกลวงเสมอ แต่ว่ามันคือ “กลยุทธ์ด้านการตลาด” เสมือนพวกเราไปเดินห้างแล้วมีพนักงานยื่นของหวานชิ้นเล็กๆให้ลองฟรีนั่นแหละขอรับ ถ้าเราชิมแล้วอร่อย (เล่นแล้วบันเทิงใจ) เราก็อยากจะอุดหนุนต่อ
slotxo การแจกเครดิตฟรีไม่ใช่การหลอกลวงเสมอ แต่ว่ามันคือ “กลยุทธ์ด้านการตลาด” เสมือนพวกเราไปเดินห้างแล้วมีพนักงานยื่นของหวานชิ้นเล็กๆให้ลองฟรีนั่นแหละขอรับ ถ้าเราชิมแล้วอร่อย (เล่นแล้วบันเทิงใจ) เราก็อยากจะอุดหนุนต่อ
 • เข้าเกมไว พอใจตั้งแต่เริ่ม: คนเล่นมือถือจำนวนมากจะถูกใจใช้งานที่เร็วทันใจอยู่แล้ว หากเข้าเว็บแล้วต้องโหลดนานๆหรือหาอะไรก็ไม่เจอ บอกเลยว่ามีสะดุดกันแน่นอน แม้กระนั้นเว็บตรงที่ดีจะจัดทุกๆสิ่งทุกๆอย่างแจ่มชัด เข้าเกมง่าย ใช้งานได้ทันที
• เข้าเกมไว พอใจตั้งแต่เริ่ม: คนเล่นมือถือจำนวนมากจะถูกใจใช้งานที่เร็วทันใจอยู่แล้ว หากเข้าเว็บแล้วต้องโหลดนานๆหรือหาอะไรก็ไม่เจอ บอกเลยว่ามีสะดุดกันแน่นอน แม้กระนั้นเว็บตรงที่ดีจะจัดทุกๆสิ่งทุกๆอย่างแจ่มชัด เข้าเกมง่าย ใช้งานได้ทันที • joker123 เว็บไซต์มองสะอาดแล้วก็อ่านง่าย: เวลาทั้งหมดทุกอย่างบนเว็บมีมากเกินไป พวกเราจะรู้สึกอ่อนล้าโดยไม่ทันรู้ตัวเลยขอรับ เว็บไซต์ที่จัดสบายตา อวดเก่งมูลไม่เกลื่อนกลาด จะทำให้พวกเรารู้สึกผ่อนคลายเวลาใช้งานมากกว่า ยิ่งถ้าหากเล่นบนโทรศัพท์มือถือ เว็บแบบงี้ยิ่งจำเป็นต้องเลยล่ะนะครับ
• joker123 เว็บไซต์มองสะอาดแล้วก็อ่านง่าย: เวลาทั้งหมดทุกอย่างบนเว็บมีมากเกินไป พวกเราจะรู้สึกอ่อนล้าโดยไม่ทันรู้ตัวเลยขอรับ เว็บไซต์ที่จัดสบายตา อวดเก่งมูลไม่เกลื่อนกลาด จะทำให้พวกเรารู้สึกผ่อนคลายเวลาใช้งานมากกว่า ยิ่งถ้าหากเล่นบนโทรศัพท์มือถือ เว็บแบบงี้ยิ่งจำเป็นต้องเลยล่ะนะครับ ขาประจำของเว็บไซต์หนึ่งได้นั้น มันก็คือการผลิตความรู้สึกว่าเว็บไซต์นี้คือของคุณ เป็นพื้นที่ของคุณ ไม่ต้องฝ่าฝืนตนเองตอนเล่นเลย
ขาประจำของเว็บไซต์หนึ่งได้นั้น มันก็คือการผลิตความรู้สึกว่าเว็บไซต์นี้คือของคุณ เป็นพื้นที่ของคุณ ไม่ต้องฝ่าฝืนตนเองตอนเล่นเลย พุซซี่888 เล่นเว็บไซต์สล็อตแตกจริงได้เงินจริง เว็บสล็อตแตกบ่อยครั้ง 2026
พุซซี่888 เล่นเว็บไซต์สล็อตแตกจริงได้เงินจริง เว็บสล็อตแตกบ่อยครั้ง 2026 พุซซี่888 ปั่นสล็อตให้ได้เงินจริง ทดสอบเล่นสล็อตฟรีได้วันแล้ววันเล่า
พุซซี่888 ปั่นสล็อตให้ได้เงินจริง ทดสอบเล่นสล็อตฟรีได้วันแล้ววันเล่า
 ฟีพบร์หลักของเกมหลายครั้งขึ้น รู้เรื่องระบบการจ่ายเงิน
ฟีพบร์หลักของเกมหลายครั้งขึ้น รู้เรื่องระบบการจ่ายเงิน 4. รับโบนัสเครดิตฟรี
4. รับโบนัสเครดิตฟรี
 918kissme ระบบการจัดการที่ “เซียน” มั่นอกมั่นใจ
918kissme ระบบการจัดการที่ “เซียน” มั่นอกมั่นใจ • กติกาขวานผ่าซาก:
• กติกาขวานผ่าซาก: 
 คนเล่นสล็อตกับ pg718 เลือกเกมจากอะไรเป็นอย่างแรกกันบ้าง?
คนเล่นสล็อตกับ pg718 เลือกเกมจากอะไรเป็นอย่างแรกกันบ้าง? ในตอนที่เปิดหน้าเกม pg718 ขึ้นมา คุณใช้เวลาเลือกเกมนานไหมนะครับ? คนโดยมากอาจจะไม่ได้ใช้เวลาเลือกเกมนานขนาดนั้น บางคนเห็นปกเกมไม่กี่วินาทีก็กดเข้าเล่นแล้ว ต่อให้ไม่รู้เรื่องว่าเกมนั้นเป็นเกมแบบไหนก็ตาม โน่นก็บอกให้เห็นเลยครับว่า การเลือกเกม pg718 ของคนเล่นสล็อตไม่ได้มาจากเหตุผลเชิงเคล็ดลับเสมอไป แม้กระนั้นมันเริ่มจากความรู้สึกแรกที่เกิดขึ้นตอนเห็นเกมสล็อตต่างหาก ในบทความนี้ ผมจะพาคุณมาดูเลยว่า อะไรส่งผลกับการตัดสินใจเลือกเกมบ้าง ถ้าเกิดคุณพร้อมและก็มาลุยกันเลย!
ในตอนที่เปิดหน้าเกม pg718 ขึ้นมา คุณใช้เวลาเลือกเกมนานไหมนะครับ? คนโดยมากอาจจะไม่ได้ใช้เวลาเลือกเกมนานขนาดนั้น บางคนเห็นปกเกมไม่กี่วินาทีก็กดเข้าเล่นแล้ว ต่อให้ไม่รู้เรื่องว่าเกมนั้นเป็นเกมแบบไหนก็ตาม โน่นก็บอกให้เห็นเลยครับว่า การเลือกเกม pg718 ของคนเล่นสล็อตไม่ได้มาจากเหตุผลเชิงเคล็ดลับเสมอไป แม้กระนั้นมันเริ่มจากความรู้สึกแรกที่เกิดขึ้นตอนเห็นเกมสล็อตต่างหาก ในบทความนี้ ผมจะพาคุณมาดูเลยว่า อะไรส่งผลกับการตัดสินใจเลือกเกมบ้าง ถ้าเกิดคุณพร้อมและก็มาลุยกันเลย! อะไรคือสิ่งแรกที่เราจะใช้ตกลงใจว่า เกม pgslot718 เกมนี้น่าเล่น?
อะไรคือสิ่งแรกที่เราจะใช้ตกลงใจว่า เกม pgslot718 เกมนี้น่าเล่น?
 สล็อตเครดิตฟรี โบนัสแตกกระจาย สายปั่นตัวจริงห้ามพลาด อิสระด้านการเงิน
สล็อตเครดิตฟรี โบนัสแตกกระจาย สายปั่นตัวจริงห้ามพลาด อิสระด้านการเงิน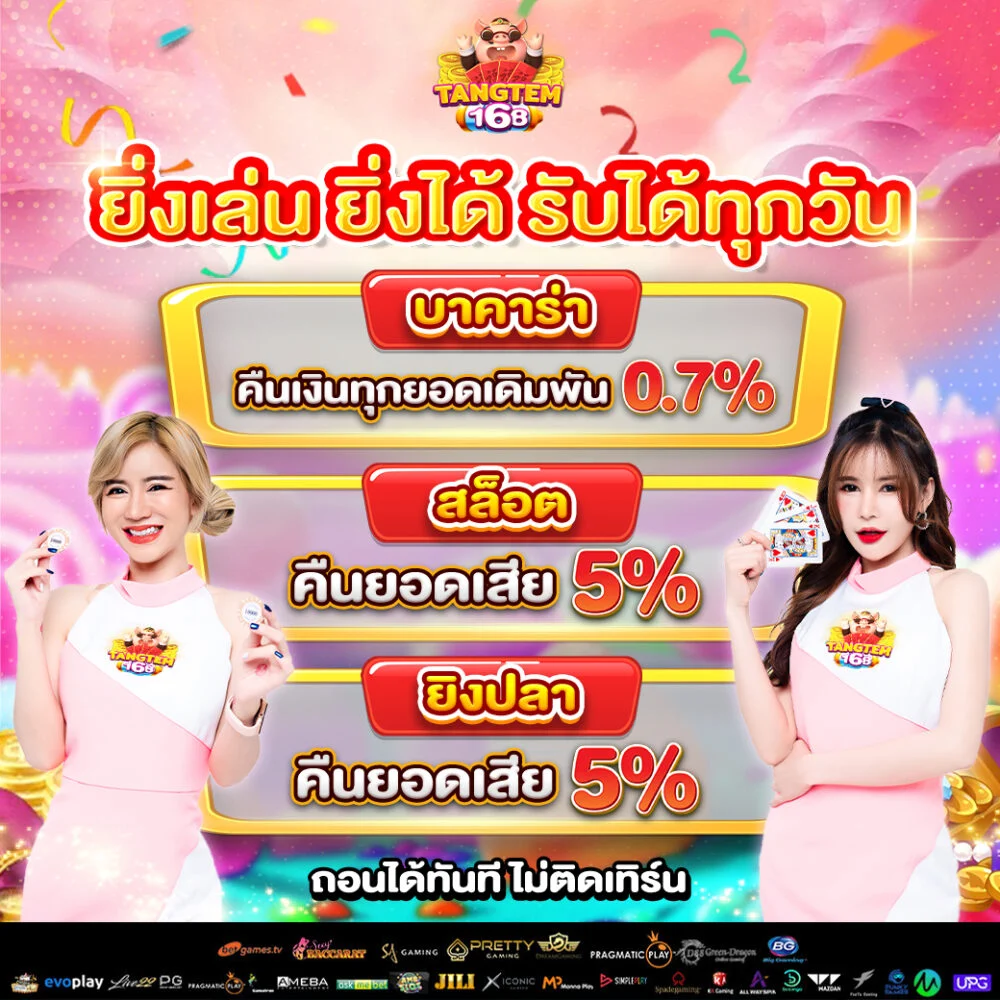 ทั้งเว็บไซต์เอเย่นต์ เว็บไซต์ผี หรือเว็บที่ประชาสัมพันธ์ว่า “แตกหนัก” แต่เพียงพอเล่นจริงกลับ “โดนกิน” จนกระทั่งเรียบ แต่ว่าจุดแปลงของผมเกิดขึ้นเมื่อเพื่อนซี้คนหนึ่งส่งลิงก์ สล็อตเครดิตฟรี มาให้ พร้อมกำชับว่า “เอ็งทดลองดู อันนี้ของแท้”
ทั้งเว็บไซต์เอเย่นต์ เว็บไซต์ผี หรือเว็บที่ประชาสัมพันธ์ว่า “แตกหนัก” แต่เพียงพอเล่นจริงกลับ “โดนกิน” จนกระทั่งเรียบ แต่ว่าจุดแปลงของผมเกิดขึ้นเมื่อเพื่อนซี้คนหนึ่งส่งลิงก์ สล็อตเครดิตฟรี มาให้ พร้อมกำชับว่า “เอ็งทดลองดู อันนี้ของแท้” ขอขอบพระคุณเว็ปไซต์
ขอขอบพระคุณเว็ปไซต์  เพราะอะไรการหยุดเล่น pg77 เป็นถึงกลายเป็นประเด็นหลักเดี๋ยวนี้?
เพราะอะไรการหยุดเล่น pg77 เป็นถึงกลายเป็นประเด็นหลักเดี๋ยวนี้?

 ถ้าจะสอบถามหา นัมเบอร์วัน ในแวดวงสล็อตออนไลน์นาทีนี้ อาจไม่มีผู้ใดกล้าไม่ยอมรับชื่อของ 188betth จริงไหมค่ะ? เพราะนี่เป็นศูนย์รวมความรื่นเริงใจที่คนไทยเลือกใช้บริการมากที่สุด ไม่ใช่แค่เนื่องจากว่าชื่อเสียงที่สั่งสมมานาน แต่เนื่องจากความ ใจถึง สำหรับในการจ่ายรางวัลรวมทั้งความเสถียรของระบบที่หาคนใดกันแน่เทียบเคียงยาก วันนี้เราจะพาทุกคนไปส่องความปังว่าเพราะเหตุไร 188betth เว็บไซต์แห่งนี้ถึงแปลงเป็นบ้านหลังใหญ่ของนักเสี่ยงโชคทั้งประเทศ!
ถ้าจะสอบถามหา นัมเบอร์วัน ในแวดวงสล็อตออนไลน์นาทีนี้ อาจไม่มีผู้ใดกล้าไม่ยอมรับชื่อของ 188betth จริงไหมค่ะ? เพราะนี่เป็นศูนย์รวมความรื่นเริงใจที่คนไทยเลือกใช้บริการมากที่สุด ไม่ใช่แค่เนื่องจากว่าชื่อเสียงที่สั่งสมมานาน แต่เนื่องจากความ ใจถึง สำหรับในการจ่ายรางวัลรวมทั้งความเสถียรของระบบที่หาคนใดกันแน่เทียบเคียงยาก วันนี้เราจะพาทุกคนไปส่องความปังว่าเพราะเหตุไร 188betth เว็บไซต์แห่งนี้ถึงแปลงเป็นบ้านหลังใหญ่ของนักเสี่ยงโชคทั้งประเทศ! เพราะอะไรคนประเทศไทยถึงทุ่มเทใจให้ตรงนี้? คำตอบง่ายๆคือความครบเครื่องค่ะ 188bet มิได้เพียงแค่เอาเกมมาลงไว้ให้เล่นเฉยๆแม้กระนั้นเขาคัดสรรเฉพาะ เกมระดับพรีเมียม ที่มีอัตราการจ่ายสูงลิบลิ่ว กราฟิกอลังการ รวมทั้งที่สำคัญเป็นจำต้อง “เล่นง่าย” ไม่สลับซับซ้อน เหมาะสมกับไลฟ์สไตล์คนสมัยใหม่ที่ชอบความเร็วและก็ความคุ้มค่า ใครกันแน่ที่เบื่อกับการเล่นเว็บไซต์เล็กๆที่มีเกมให้เลือกน้อย หรือถอนเงินทีไรต้องลุ้นตัวโก่ง บอกเลยว่ามาจอดป้ายตรงนี้คือจบทุกความอยาก จัดแจงให้พร้อมนะคะ เพราะพวกเรากำลังจะพาคุณไปพบกับ 188betth เส้นทางคนมั่งคั่งที่สร้างได้อย่างง่ายดายแค่ปลายนิ้วสัมผัส!
เพราะอะไรคนประเทศไทยถึงทุ่มเทใจให้ตรงนี้? คำตอบง่ายๆคือความครบเครื่องค่ะ 188bet มิได้เพียงแค่เอาเกมมาลงไว้ให้เล่นเฉยๆแม้กระนั้นเขาคัดสรรเฉพาะ เกมระดับพรีเมียม ที่มีอัตราการจ่ายสูงลิบลิ่ว กราฟิกอลังการ รวมทั้งที่สำคัญเป็นจำต้อง “เล่นง่าย” ไม่สลับซับซ้อน เหมาะสมกับไลฟ์สไตล์คนสมัยใหม่ที่ชอบความเร็วและก็ความคุ้มค่า ใครกันแน่ที่เบื่อกับการเล่นเว็บไซต์เล็กๆที่มีเกมให้เลือกน้อย หรือถอนเงินทีไรต้องลุ้นตัวโก่ง บอกเลยว่ามาจอดป้ายตรงนี้คือจบทุกความอยาก จัดแจงให้พร้อมนะคะ เพราะพวกเรากำลังจะพาคุณไปพบกับ 188betth เส้นทางคนมั่งคั่งที่สร้างได้อย่างง่ายดายแค่ปลายนิ้วสัมผัส!

 สล็อต เล่นง่าย แม้กระนั้นทำเงินให้เป็นประจำไม่ง่ายเหมือนอย่างที่คิด เพราะอะไร?
สล็อต เล่นง่าย แม้กระนั้นทำเงินให้เป็นประจำไม่ง่ายเหมือนอย่างที่คิด เพราะอะไร?

 คนส่วนมากเล่น joker123 แล้วพลาดตรงไหนบ้าง?
คนส่วนมากเล่น joker123 แล้วพลาดตรงไหนบ้าง? ถ้าคุณเคยเล่น
ถ้าคุณเคยเล่น  คุณกำลังเล่น โจ๊กเกอร์123 พลาดโดยไม่ทันรู้ตัวรึเปล่า? มาหาคำตอบกัน!
คุณกำลังเล่น โจ๊กเกอร์123 พลาดโดยไม่ทันรู้ตัวรึเปล่า? มาหาคำตอบกัน!
 หลังจากฝากเงิน ระบบก็อัปเดตยอดค่อนข้างจะไว ผมไม่ต้องรอนานเหมือนบางเว็บไซต์ที่เคยใช้มาก่อน ต่อจากนั้นก็ทดลองเข้าเล่นสล็อตทันที สิ่งที่รู้สึกได้เป็นความลื่นไถลของระบบ เกมโหลดเร็ว แปลงเกมก็ไม่สะดุด ทำให้เล่นต่อเนื่องได้แบบไม่เสียอารมณ์
หลังจากฝากเงิน ระบบก็อัปเดตยอดค่อนข้างจะไว ผมไม่ต้องรอนานเหมือนบางเว็บไซต์ที่เคยใช้มาก่อน ต่อจากนั้นก็ทดลองเข้าเล่นสล็อตทันที สิ่งที่รู้สึกได้เป็นความลื่นไถลของระบบ เกมโหลดเร็ว แปลงเกมก็ไม่สะดุด ทำให้เล่นต่อเนื่องได้แบบไม่เสียอารมณ์

 เว็บตรงแบบ 918kissauto เปลี่ยนความประพฤติการเล่นของผู้เล่นได้อย่างไร?
เว็บตรงแบบ 918kissauto เปลี่ยนความประพฤติการเล่นของผู้เล่นได้อย่างไร? เมื่อระบบของ 918kiss รวดเร็วและสม่ำเสมอ ผู้เล่นดีกว่าเรื่องอะไรบ้าง?
เมื่อระบบของ 918kiss รวดเร็วและสม่ำเสมอ ผู้เล่นดีกว่าเรื่องอะไรบ้าง?
 ตอนต้นผมมิได้คิดว่าจะมาพบกับอะไรอย่างนี้เลยจ้า กับเว็บไซต์อย่าง scr888 ที่หลายคนพูดกันว่า “สล็อตแตกง่าย” ในตอนนั้นก็รู้สึกว่าเป็นเพียงแค่คำโฆษณาทั่วๆไป แม้กระนั้นพอลองเล่นจริงๆบอกเลยว่ามันมีอะไรแปลกๆแบบงงมากๆเกิดขึ้นจริง
ตอนต้นผมมิได้คิดว่าจะมาพบกับอะไรอย่างนี้เลยจ้า กับเว็บไซต์อย่าง scr888 ที่หลายคนพูดกันว่า “สล็อตแตกง่าย” ในตอนนั้นก็รู้สึกว่าเป็นเพียงแค่คำโฆษณาทั่วๆไป แม้กระนั้นพอลองเล่นจริงๆบอกเลยว่ามันมีอะไรแปลกๆแบบงงมากๆเกิดขึ้นจริง สิ่งที่กระจ่างมากมายคือ “จังหวะเกมเร็ว” เป็นไม่ต้องรอนานให้เสียอารมณ์ เหมาะสมกับคนที่เกลียดชังนั่งเฝ้าหน้าจอแบบลุ้นยาวๆอยากได้อะไรไวๆเล่นแล้วมีอะไรให้ตื่นเต้นตลอดระยะเวลา ยิ่งเล่นบนโทรศัพท์มือถือยิ่งสนุก เนื่องจากว่าระบบลื่น กดหมุนตลอดได้แบบไม่มีสะดุด
สิ่งที่กระจ่างมากมายคือ “จังหวะเกมเร็ว” เป็นไม่ต้องรอนานให้เสียอารมณ์ เหมาะสมกับคนที่เกลียดชังนั่งเฝ้าหน้าจอแบบลุ้นยาวๆอยากได้อะไรไวๆเล่นแล้วมีอะไรให้ตื่นเต้นตลอดระยะเวลา ยิ่งเล่นบนโทรศัพท์มือถือยิ่งสนุก เนื่องจากว่าระบบลื่น กดหมุนตลอดได้แบบไม่มีสะดุด แล้วจุดพีคมันอยู่ตอนที่โบนัสเข้าแบบเข้าแผนไม่ทันตั้งตัวอีกแล้ว ราว 20 กว่าตา Scatter ลงครบ เข้าโหมดฟรีสปินทันที รอบนั้นคือได้ตลอดตัวคูณและก็เครื่องหมายพิเศษออกต่อเนื่อง ยอดเงินพุ่งขึ้นแบบเห็นชัด จากทุนไม่มากมาย แปลงเป็นกำไรหลักร้อยได้ในเวลาไม่นาน
แล้วจุดพีคมันอยู่ตอนที่โบนัสเข้าแบบเข้าแผนไม่ทันตั้งตัวอีกแล้ว ราว 20 กว่าตา Scatter ลงครบ เข้าโหมดฟรีสปินทันที รอบนั้นคือได้ตลอดตัวคูณและก็เครื่องหมายพิเศษออกต่อเนื่อง ยอดเงินพุ่งขึ้นแบบเห็นชัด จากทุนไม่มากมาย แปลงเป็นกำไรหลักร้อยได้ในเวลาไม่นาน
 ผมรู้จักกับ สล็อต168 จากการที่เพื่อนฝูงเสนอแนะในช่วงที่กำลังมองหา “สล็อตเว็บไซต์ตรง” ที่น่าไว้วางใจ เล่นง่าย รวมทั้งที่สำคัญเป็นไม่ต้องมีขั้นตอนยุ่งยาก ตอนต้นก็ยังลังเลอยู่แบบเดียวกัน ด้วยเหตุว่าตอนนี้เว็บไซต์สล็อตมีให้เลือกมากไม่น้อยเลยทีเดียว แต่ว่าพอใช้ได้ทดลองใช้งานจริง บอกเลยว่าประสบการณ์ที่ได้รับมันต่างออกไปจริงๆ
ผมรู้จักกับ สล็อต168 จากการที่เพื่อนฝูงเสนอแนะในช่วงที่กำลังมองหา “สล็อตเว็บไซต์ตรง” ที่น่าไว้วางใจ เล่นง่าย รวมทั้งที่สำคัญเป็นไม่ต้องมีขั้นตอนยุ่งยาก ตอนต้นก็ยังลังเลอยู่แบบเดียวกัน ด้วยเหตุว่าตอนนี้เว็บไซต์สล็อตมีให้เลือกมากไม่น้อยเลยทีเดียว แต่ว่าพอใช้ได้ทดลองใช้งานจริง บอกเลยว่าประสบการณ์ที่ได้รับมันต่างออกไปจริงๆ “ระบบฝาก-ถอน” ที่รวดเร็วทันใจมาก แบบที่สามารถพูดได้ว่าไม่ต้องรอนานเลย ฝากเงินเข้าไปไม่ถึงไม่กี่วินาทีก็สามารถเข้าเล่นเกมได้โดยทันที ส่วนตอนถอนเงินก็เช่นกัน เคยลองถอนยามดึกดื่นๆก็ยังได้รับเงินเร็ว ไม่มีค้าง ไม่มีเลื่อน ทำให้มีความรู้สึกแน่ใจว่าเว็บไซต์แห่งนี้มีระบบออโต้ที่เสถียรจริงๆ
“ระบบฝาก-ถอน” ที่รวดเร็วทันใจมาก แบบที่สามารถพูดได้ว่าไม่ต้องรอนานเลย ฝากเงินเข้าไปไม่ถึงไม่กี่วินาทีก็สามารถเข้าเล่นเกมได้โดยทันที ส่วนตอนถอนเงินก็เช่นกัน เคยลองถอนยามดึกดื่นๆก็ยังได้รับเงินเร็ว ไม่มีค้าง ไม่มีเลื่อน ทำให้มีความรู้สึกแน่ใจว่าเว็บไซต์แห่งนี้มีระบบออโต้ที่เสถียรจริงๆ


 4. รับโบนัสเครดิตฟรี
4. รับโบนัสเครดิตฟรี
 ถ้าเกิดถามว่าอะไรที่ทำให้ เศรษฐี99 สะดุดตาออกมาจากเว็บไซต์พนันออนไลน์นับพัน บอกเลยว่าไม่ใช่แค่เรื่องชื่อที่เป็นมงคล (แต่ชื่อก็ปังจริงนะ คนรวยเชียวนะคุณ!) แต่มันเป็นส่วนประกอบความเลิศเลอรอบด้านที่เขาจัดมาให้แบบจุกๆดังนี้จ้ะ:
ถ้าเกิดถามว่าอะไรที่ทำให้ เศรษฐี99 สะดุดตาออกมาจากเว็บไซต์พนันออนไลน์นับพัน บอกเลยว่าไม่ใช่แค่เรื่องชื่อที่เป็นมงคล (แต่ชื่อก็ปังจริงนะ คนรวยเชียวนะคุณ!) แต่มันเป็นส่วนประกอบความเลิศเลอรอบด้านที่เขาจัดมาให้แบบจุกๆดังนี้จ้ะ: sretthi99 แจกโปรเด็ด โปรฉ่ำ ที่สายเอาคุ้มต้องกรี๊ด! คุ้มสุดๆห้ามพลาด !
sretthi99 แจกโปรเด็ด โปรฉ่ำ ที่สายเอาคุ้มต้องกรี๊ด! คุ้มสุดๆห้ามพลาด ! จบท้ายด้วย โปรโมชั่นชี้แนะสหาย ที่ทำเอาคนไม่ใช่น้อยกลายเป็นคนรวยตัวน้อยไปแล้ว แค่แชร์ลิงก์ให้เพื่อนพ้องมาจอยความสนุกสนานร่าเริงร่วมกัน คุณก็รอรับค่าคอมมิชชั่นแบบ Passive Income ได้เลย ยิ่งมิตรสหายเยอะ คุณยิ่งได้มากมาย กินเปอร์เซ็นต์กันยาวๆแบบไม่ต้องลงทุนเองสักบาทเดียว บอกเลยว่าโปรโมชั่นของ สล็อต99 คือการรวมทุกความคุ้มค่าไว้ภายในที่เดียว ทั้งยังแจก ทั้งยังแถม ตลอดทั้งคืนกำไร ผู้ใดกันพลาดไปบอกเลยว่าเสียดายของสุดๆจ้ะ!
จบท้ายด้วย โปรโมชั่นชี้แนะสหาย ที่ทำเอาคนไม่ใช่น้อยกลายเป็นคนรวยตัวน้อยไปแล้ว แค่แชร์ลิงก์ให้เพื่อนพ้องมาจอยความสนุกสนานร่าเริงร่วมกัน คุณก็รอรับค่าคอมมิชชั่นแบบ Passive Income ได้เลย ยิ่งมิตรสหายเยอะ คุณยิ่งได้มากมาย กินเปอร์เซ็นต์กันยาวๆแบบไม่ต้องลงทุนเองสักบาทเดียว บอกเลยว่าโปรโมชั่นของ สล็อต99 คือการรวมทุกความคุ้มค่าไว้ภายในที่เดียว ทั้งยังแจก ทั้งยังแถม ตลอดทั้งคืนกำไร ผู้ใดกันพลาดไปบอกเลยว่าเสียดายของสุดๆจ้ะ! ขอขอบคุณ Ref.
ขอขอบคุณ Ref.  กีฬาบอลเป็นที่นิยมมาอย่างยาวนานเนื่องจากว่าเป็นกีฬาที่เชียร์แล้วรู้สึกเพลิดเพลินเจริญใจโดยเฉพาะอย่างยิ่งการพนันบอล กลุ่มที่เลือกมีความใส่ใจในระบบก็จะยิ่งผลักดันสมรรถนะสำหรับเพื่อการ เว็บแทงบอล ให้มีความสุขเพิ่มยิ่งขึ้นแต่หากคุณเป็นมือใหม่แล้ว มีความรู้สึกกังวลใจสำหรับในการแทงบอลพวกเราชี้แนะว่าคุณควรเลือกเว็บแทงบอลที่มีศักยภาพสูง
กีฬาบอลเป็นที่นิยมมาอย่างยาวนานเนื่องจากว่าเป็นกีฬาที่เชียร์แล้วรู้สึกเพลิดเพลินเจริญใจโดยเฉพาะอย่างยิ่งการพนันบอล กลุ่มที่เลือกมีความใส่ใจในระบบก็จะยิ่งผลักดันสมรรถนะสำหรับเพื่อการ เว็บแทงบอล ให้มีความสุขเพิ่มยิ่งขึ้นแต่หากคุณเป็นมือใหม่แล้ว มีความรู้สึกกังวลใจสำหรับในการแทงบอลพวกเราชี้แนะว่าคุณควรเลือกเว็บแทงบอลที่มีศักยภาพสูง

 ทำไม pgslotfish ถึงเหมาะกับคนที่ “คอยเป็น” มากยิ่งกว่า “เร่งเป็น”
ทำไม pgslotfish ถึงเหมาะกับคนที่ “คอยเป็น” มากยิ่งกว่า “เร่งเป็น”

 จากมือใหม่สู่คนเล่นประจำใน sagame168th แตกต่างกันเท่าไร?
จากมือใหม่สู่คนเล่นประจำใน sagame168th แตกต่างกันเท่าไร? ผมชอบระลึกถึงวันแรกที่ผมเข้ามาเล่น sagame อยู่เสมอเลยนะครับ เพราะเหตุว่าเวลามองย้อนกลับไปแล้วมาเทียบกับตัวผมเองในปัจจุบัน ผมบอกเลยว่าผมแปรไปมากมายๆซึ่งการเปลี่ยนแปลงที่เกิดขึ้นนั้น มันไม่ใช่ว่าผมเก่งขึ้นหรือเล่นได้กำไรเยอะขึ้นเรื่อยๆนะครับ แต่มันคือเรื่องของแนวทางคิด การมองเกม หรือการจัดการกับปัญหาที่เกิดขึ้นมากกว่า ซึ่งผมเชื่อว่า คนที่เป็นมือใหม่จนกระทั่งมาเป็นคนเล่นประจำก็คงจะไม่แตกต่างจากผมแน่ๆ โดยเหตุนั้น ผมจะพามามองเลยว่า มือใหม่กับคนเล่นประจำไม่เหมือนกันแค่ไหน
ผมชอบระลึกถึงวันแรกที่ผมเข้ามาเล่น sagame อยู่เสมอเลยนะครับ เพราะเหตุว่าเวลามองย้อนกลับไปแล้วมาเทียบกับตัวผมเองในปัจจุบัน ผมบอกเลยว่าผมแปรไปมากมายๆซึ่งการเปลี่ยนแปลงที่เกิดขึ้นนั้น มันไม่ใช่ว่าผมเก่งขึ้นหรือเล่นได้กำไรเยอะขึ้นเรื่อยๆนะครับ แต่มันคือเรื่องของแนวทางคิด การมองเกม หรือการจัดการกับปัญหาที่เกิดขึ้นมากกว่า ซึ่งผมเชื่อว่า คนที่เป็นมือใหม่จนกระทั่งมาเป็นคนเล่นประจำก็คงจะไม่แตกต่างจากผมแน่ๆ โดยเหตุนั้น ผมจะพามามองเลยว่า มือใหม่กับคนเล่นประจำไม่เหมือนกันแค่ไหน
 รู้สึกได้ชัดเป็น “ความนำสมัยของแพลตฟอร์มเกม” หลายเว็บรวมเกมจากค่ายดังทั้งโลกมาไว้ภายในที่เดียว สล็อตออนไลน์ ทำให้ผู้เล่นสามารถเลือกเล่นได้นานัปการแนว ไม่ว่าจะเป็นสล็อตแนวผจญภัย แนวแฟนตาซี หรือแนวผลไม้คลาสสิกแบบดั้งเดิม แต่ละเกมจะมีฟีพบร์พิเศษไม่เหมือนกัน ดังเช่นว่า เครื่องหมายโบนัส ฟรีสปิน หรือรอบพิเศษที่เพิ่มความระทึกใจในการเล่น
รู้สึกได้ชัดเป็น “ความนำสมัยของแพลตฟอร์มเกม” หลายเว็บรวมเกมจากค่ายดังทั้งโลกมาไว้ภายในที่เดียว สล็อตออนไลน์ ทำให้ผู้เล่นสามารถเลือกเล่นได้นานัปการแนว ไม่ว่าจะเป็นสล็อตแนวผจญภัย แนวแฟนตาซี หรือแนวผลไม้คลาสสิกแบบดั้งเดิม แต่ละเกมจะมีฟีพบร์พิเศษไม่เหมือนกัน ดังเช่นว่า เครื่องหมายโบนัส ฟรีสปิน หรือรอบพิเศษที่เพิ่มความระทึกใจในการเล่น 188bet ในมุมของประสบการณ์ส่วนตัว ผมสังเกตว่าเกมยุคใหม่ย้ำ “ความเพลิดเพลินและการมีส่วนร่วม” เพิ่มมากขึ้น ไม่ใช่แค่การกดหมุนแล้วรอคอยผลเหมือนยุคเก่า แม้กระนั้นจะมีเอฟเฟกต์ภาพ เสียง และก็ระบบเลเวลที่ทำให้เกิดความรู้สึกเหมือนกำลังเล่นเกมมากกว่าเพียงแค่เสี่ยงดวงปกติ ทำให้ผู้เล่นหลายท่านรู้สึกเพลินใจรวมทั้งใช้เวลาไปกับมันได้ง่าย
188bet ในมุมของประสบการณ์ส่วนตัว ผมสังเกตว่าเกมยุคใหม่ย้ำ “ความเพลิดเพลินและการมีส่วนร่วม” เพิ่มมากขึ้น ไม่ใช่แค่การกดหมุนแล้วรอคอยผลเหมือนยุคเก่า แม้กระนั้นจะมีเอฟเฟกต์ภาพ เสียง และก็ระบบเลเวลที่ทำให้เกิดความรู้สึกเหมือนกำลังเล่นเกมมากกว่าเพียงแค่เสี่ยงดวงปกติ ทำให้ผู้เล่นหลายท่านรู้สึกเพลินใจรวมทั้งใช้เวลาไปกับมันได้ง่าย อีกสิ่งที่พบเป็นเรื่องของ “ความสบายสำหรับเพื่อการเข้าถึง” ปัจจุบันหลายแพลตฟอร์มรองรับโทรศัพท์มือถือเต็มรูปแบบ ทำให้สามารถเล่นได้ทุกที่ที่ต้องการ ไม่ว่าจะอยู่บ้าน หรือระหว่างเดินทาง ก็สามารถเข้าไปทดลองเล่นเกมใหม่ๆได้ตลอด ซึ่งถือได้ว่าข้อดีสำคัญของสมัยดิจิทัล
อีกสิ่งที่พบเป็นเรื่องของ “ความสบายสำหรับเพื่อการเข้าถึง” ปัจจุบันหลายแพลตฟอร์มรองรับโทรศัพท์มือถือเต็มรูปแบบ ทำให้สามารถเล่นได้ทุกที่ที่ต้องการ ไม่ว่าจะอยู่บ้าน หรือระหว่างเดินทาง ก็สามารถเข้าไปทดลองเล่นเกมใหม่ๆได้ตลอด ซึ่งถือได้ว่าข้อดีสำคัญของสมัยดิจิทัล
 เดี๋ยวนี้ผมสังเกตว่ากระแสเกมสล็อตออนไลน์ยังคงมาแรงแบบไม่มีตก ไม่ว่าจะเลื่อนดูโซเชียลหรือคุยกับเพื่อนพ้องก็ชอบได้ยินคำว่า สล็อต pgslot แตกง่าย หรือเว็บไซต์ตรงเล่นแล้วได้เงินจริงอยู่ตลอด ซึ่งจากที่ผมเองก็ทดลองเล่นมาหลายเว็บไซต์ จำต้องยอมรับเลยว่าการเลือกเว็บเป็นสิ่งจำเป็นมาก เพราะว่าถ้าเกิดเลือกไม่ถูก นอกจากจะเสียเวลาแล้ว ยังเสี่ยงเสียเงินเสียทองแบบไม่คุ้มอีกด้วย
เดี๋ยวนี้ผมสังเกตว่ากระแสเกมสล็อตออนไลน์ยังคงมาแรงแบบไม่มีตก ไม่ว่าจะเลื่อนดูโซเชียลหรือคุยกับเพื่อนพ้องก็ชอบได้ยินคำว่า สล็อต pgslot แตกง่าย หรือเว็บไซต์ตรงเล่นแล้วได้เงินจริงอยู่ตลอด ซึ่งจากที่ผมเองก็ทดลองเล่นมาหลายเว็บไซต์ จำต้องยอมรับเลยว่าการเลือกเว็บเป็นสิ่งจำเป็นมาก เพราะว่าถ้าเกิดเลือกไม่ถูก นอกจากจะเสียเวลาแล้ว ยังเสี่ยงเสียเงินเสียทองแบบไม่คุ้มอีกด้วย ตราบจนกระทั่งผมได้มาเจอกับ pgslot สล็อตเว็บตรง
ตราบจนกระทั่งผมได้มาเจอกับ pgslot สล็อตเว็บตรง  funny18 สล็อตเว็บตรง ใช้งานจริงแล้วรู้สึกว่าเป็นมากกว่าเพียงแค่เว็บสล็อตทั่วไป
funny18 สล็อตเว็บตรง ใช้งานจริงแล้วรู้สึกว่าเป็นมากกว่าเพียงแค่เว็บสล็อตทั่วไป ภายหลังที่ผมได้ลองใช้งาน funny18 มาสักระยะหนึ่ง ต้องบอกเลยว่าเป็นอีกหนึ่งเว็บไซต์ที่ทำออกมาได้ค่อนข้างจะดีในหลายๆด้าน ทั้งยังเรื่องของระบบ ความเสถียรภาพ และก็ประสิทธิภาพของเกม
ภายหลังที่ผมได้ลองใช้งาน funny18 มาสักระยะหนึ่ง ต้องบอกเลยว่าเป็นอีกหนึ่งเว็บไซต์ที่ทำออกมาได้ค่อนข้างจะดีในหลายๆด้าน ทั้งยังเรื่องของระบบ ความเสถียรภาพ และก็ประสิทธิภาพของเกม มาร่วมสนุกกับ เกมสล็อตนานัปการรวมทั้งโปรโมชั่นล้นหลามวันนี้ พร้อมรับสิทธิพิเศษ และก็ประสบการณ์การเล่นที่ไม่ซ้ำใคร
มาร่วมสนุกกับ เกมสล็อตนานัปการรวมทั้งโปรโมชั่นล้นหลามวันนี้ พร้อมรับสิทธิพิเศษ และก็ประสบการณ์การเล่นที่ไม่ซ้ำใคร ลักษณะเด่นก็คือเว็บไซต์นี้ มีระบบระเบียบทดลองเล่นฟรี ผู้เล่นสามารถเข้าไปทดลองหมุนสล็อตทุกแบบอย่างโดยไม่ต้องเพิ่มเงินจริงก่อน ซึ่งเหมาะสำหรับอีกทั้งมือใหม่ที่เพิ่งเริ่มรวมทั้งผู้เล่นที่ต้องการทำความเข้าใจฟีเจอร์ของเกมต่างๆก่อนเดิมพันจริง
ลักษณะเด่นก็คือเว็บไซต์นี้ มีระบบระเบียบทดลองเล่นฟรี ผู้เล่นสามารถเข้าไปทดลองหมุนสล็อตทุกแบบอย่างโดยไม่ต้องเพิ่มเงินจริงก่อน ซึ่งเหมาะสำหรับอีกทั้งมือใหม่ที่เพิ่งเริ่มรวมทั้งผู้เล่นที่ต้องการทำความเข้าใจฟีเจอร์ของเกมต่างๆก่อนเดิมพันจริง (หมายเหตุ: ตัวอย่างเกมกลุ่มนี้เป็นแถวทางตามแนวโน้มสล็อตยอดฮิต และอาจมีการเปลี่ยนแปลงตามแต่ละเว็บเอง)
(หมายเหตุ: ตัวอย่างเกมกลุ่มนี้เป็นแถวทางตามแนวโน้มสล็อตยอดฮิต และอาจมีการเปลี่ยนแปลงตามแต่ละเว็บเอง) เล่น
เล่น 2. กฎการจั่วทำให้ Banker เหนือกว่ามากยิ่งกว่า
2. กฎการจั่วทำให้ Banker เหนือกว่ามากยิ่งกว่า

 ตอนแรกผมรู้จัก สล็อต168 จากเพื่อนที่แนะนำมา เขากล่าวว่าเป็นเว็บที่รวมสล็อตหลายค่ายที่ได้รับความนิยม อย่างเช่น PG, Joker,
ตอนแรกผมรู้จัก สล็อต168 จากเพื่อนที่แนะนำมา เขากล่าวว่าเป็นเว็บที่รวมสล็อตหลายค่ายที่ได้รับความนิยม อย่างเช่น PG, Joker,  สิ่งที่ผมชอบสูงที่สุดคือ สามารถเล่นได้ทุกค่ายในเว็บไซต์เดียว ไม่ต้องเสียเวลาไปสมัครหลายเว็บไซต์เหมือนครั้งก่อน ปกติผมจะชอบเล่นสล็อตของหลายค่าย เพราะแต่ละค่ายก็มีลักษณะเด่นต่างกัน อาทิเช่น บางเกมโบนัสแตกง่าย บางเกมแจ็คพอตใหญ่ ทำให้การเล่นไม่น่าเบื่อ
สิ่งที่ผมชอบสูงที่สุดคือ สามารถเล่นได้ทุกค่ายในเว็บไซต์เดียว ไม่ต้องเสียเวลาไปสมัครหลายเว็บไซต์เหมือนครั้งก่อน ปกติผมจะชอบเล่นสล็อตของหลายค่าย เพราะแต่ละค่ายก็มีลักษณะเด่นต่างกัน อาทิเช่น บางเกมโบนัสแตกง่าย บางเกมแจ็คพอตใหญ่ ทำให้การเล่นไม่น่าเบื่อ

 pussy888 กับการฝึกฝนสังเกตเกมด้วยตัวเองแบบง่ายๆ
pussy888 กับการฝึกฝนสังเกตเกมด้วยตัวเองแบบง่ายๆ คนไม่ใช่น้อยจะเข้าใจว่า การเล่น pussy888play นั้นจำเป็นต้องพึ่งพิงสูตรหรือเคล็ดลับต่างๆที่ซับซ้อนเพียงแค่นั้นถึงจะเล่นได้ดิบได้ดี แต่พอใช้ได้เล่นด้วยตัวเองไปครู่หนึ่ง คุณจะพบว่าเรื่องจริงแล้ว สูตรหรือเคล็ดลับอะไรพวกนั้นมันไม่จำเป็นเลยครับ ด้วยเหตุว่าคุณเองสามารถพิจารณาด้วยตัวเองได้ ซึ่งเป็นความสามารถการเล่น pussy888play ที่สำคัญกว่ามาก มันจะทำให้คุณเข้าใจเกมได้มากกว่า ไม่ใช่แค่ปฏิบัติตามบุคคลอื่นเท่านั้น ในเนื้อหานี้ ผมจะพาคุณมาดูกระบวนการฝึกฝนดูด้วยตัวเองแบบง่ายๆที่ไม่ต้องมีพื้นฐานก็เริ่มได้รวมทั้งใช้ได้จริงแน่นอนครับผม
คนไม่ใช่น้อยจะเข้าใจว่า การเล่น pussy888play นั้นจำเป็นต้องพึ่งพิงสูตรหรือเคล็ดลับต่างๆที่ซับซ้อนเพียงแค่นั้นถึงจะเล่นได้ดิบได้ดี แต่พอใช้ได้เล่นด้วยตัวเองไปครู่หนึ่ง คุณจะพบว่าเรื่องจริงแล้ว สูตรหรือเคล็ดลับอะไรพวกนั้นมันไม่จำเป็นเลยครับ ด้วยเหตุว่าคุณเองสามารถพิจารณาด้วยตัวเองได้ ซึ่งเป็นความสามารถการเล่น pussy888play ที่สำคัญกว่ามาก มันจะทำให้คุณเข้าใจเกมได้มากกว่า ไม่ใช่แค่ปฏิบัติตามบุคคลอื่นเท่านั้น ในเนื้อหานี้ ผมจะพาคุณมาดูกระบวนการฝึกฝนดูด้วยตัวเองแบบง่ายๆที่ไม่ต้องมีพื้นฐานก็เริ่มได้รวมทั้งใช้ได้จริงแน่นอนครับผม การฝึกหัดดูเกม pussy888 ไม่ได้มาจากผู้กระทำดเล่นจำนวนไม่ใช่น้อยแค่นั้นนะครับ เพราะเหตุว่าถ้าคุณกดเล่นเยอะมากๆแต่ว่าเล่นไปเรื่อยๆแบบไม่ได้พินิจอะไรก็ไม่ถือว่าเป็นการฝึก คุณจะต้องเริ่มจากการดูให้รู้เรื่องก่อนว่าเกมกำลังเป็นไปแบบไหนด้วย
การฝึกหัดดูเกม pussy888 ไม่ได้มาจากผู้กระทำดเล่นจำนวนไม่ใช่น้อยแค่นั้นนะครับ เพราะเหตุว่าถ้าคุณกดเล่นเยอะมากๆแต่ว่าเล่นไปเรื่อยๆแบบไม่ได้พินิจอะไรก็ไม่ถือว่าเป็นการฝึก คุณจะต้องเริ่มจากการดูให้รู้เรื่องก่อนว่าเกมกำลังเป็นไปแบบไหนด้วย
 อะไรเป็นเหตุผลจริงๆที่ทำให้เลือก sagame168 กันแน่? หากพวกเราตัดเรื่องโฆษณาออกไป คนที่เล่นบาคาร่าจริงจะเลือกเว็บไซต์จากความรู้สึกตอนใช้งานจริงนี่แหละครับ ซึ่งเว็บตรงของเราเป็นเว็บที่ตอบโจทย์สูงที่สุด เพราะเราเน้นย้ำสำหรับเพื่อการสร้างระบบที่ฐานรากก่อนเสมอ นี่ก็เลยเป็นข้อเด่นในการให้บริการ sagame168th ระยะยาว ทำให้ไม่ต้องปรับพฤติกรรมมากและเล่นได้อย่างสบายใจ
อะไรเป็นเหตุผลจริงๆที่ทำให้เลือก sagame168 กันแน่? หากพวกเราตัดเรื่องโฆษณาออกไป คนที่เล่นบาคาร่าจริงจะเลือกเว็บไซต์จากความรู้สึกตอนใช้งานจริงนี่แหละครับ ซึ่งเว็บตรงของเราเป็นเว็บที่ตอบโจทย์สูงที่สุด เพราะเราเน้นย้ำสำหรับเพื่อการสร้างระบบที่ฐานรากก่อนเสมอ นี่ก็เลยเป็นข้อเด่นในการให้บริการ sagame168th ระยะยาว ทำให้ไม่ต้องปรับพฤติกรรมมากและเล่นได้อย่างสบายใจ


 ถึงจะเริ่มมองเห็นได้ แน่นอนว่า มันไม่ใช่เรื่องที่เกี่ยวข้องกับการทำนายผลลัพธ์ของเกมได้แบบ 100% อยู่แล้ว แต่การดูความประพฤติเกมระหว่างการเล่นจะช่วยทำให้พวกเราเข้าใจเกมนั้นๆมากขึ้น ในเนื้อหานี้ ผมจะพาคุณมาอ่านจังหวะเกมแบบง่ายๆแบบมืออาชีพชี้แนะเองกับมือเลยครับผม การันตีเลยว่า อ่านเนื้อหานี้จบแล้ว อ่านจังหวะเกมต่อกันได้เลย หากคุณพร้อมและฝ่าเลยขอรับ!
ถึงจะเริ่มมองเห็นได้ แน่นอนว่า มันไม่ใช่เรื่องที่เกี่ยวข้องกับการทำนายผลลัพธ์ของเกมได้แบบ 100% อยู่แล้ว แต่การดูความประพฤติเกมระหว่างการเล่นจะช่วยทำให้พวกเราเข้าใจเกมนั้นๆมากขึ้น ในเนื้อหานี้ ผมจะพาคุณมาอ่านจังหวะเกมแบบง่ายๆแบบมืออาชีพชี้แนะเองกับมือเลยครับผม การันตีเลยว่า อ่านเนื้อหานี้จบแล้ว อ่านจังหวะเกมต่อกันได้เลย หากคุณพร้อมและฝ่าเลยขอรับ! การอ่านจังหวะเกม pgslot เป็นอย่างไร? แล้วพวกเราดูจากอะไรได้บ้าง?
การอ่านจังหวะเกม pgslot เป็นอย่างไร? แล้วพวกเราดูจากอะไรได้บ้าง?
 เพราะอะไรสายปั่นเฉยๆถึงเลือก สล็อต66 เยอะขึ้น? ผมมีเพื่อนฝูงที่เป็นสายปั่นเฉยๆเยอะมาก คนที่ไม่ได้โพสต์หรือรีวิวอะไรเลย เล่นเงียบๆของตัวเองไปเรื่อยๆแค่นั้น ทุกคนต่างเลือก
เพราะอะไรสายปั่นเฉยๆถึงเลือก สล็อต66 เยอะขึ้น? ผมมีเพื่อนฝูงที่เป็นสายปั่นเฉยๆเยอะมาก คนที่ไม่ได้โพสต์หรือรีวิวอะไรเลย เล่นเงียบๆของตัวเองไปเรื่อยๆแค่นั้น ทุกคนต่างเลือก  สายปั่นเงียบๆมองหาอะไรใน pgslot มากยิ่งกว่าความลื่นไถลไหลของเกมกันนะ?
สายปั่นเงียบๆมองหาอะไรใน pgslot มากยิ่งกว่าความลื่นไถลไหลของเกมกันนะ?
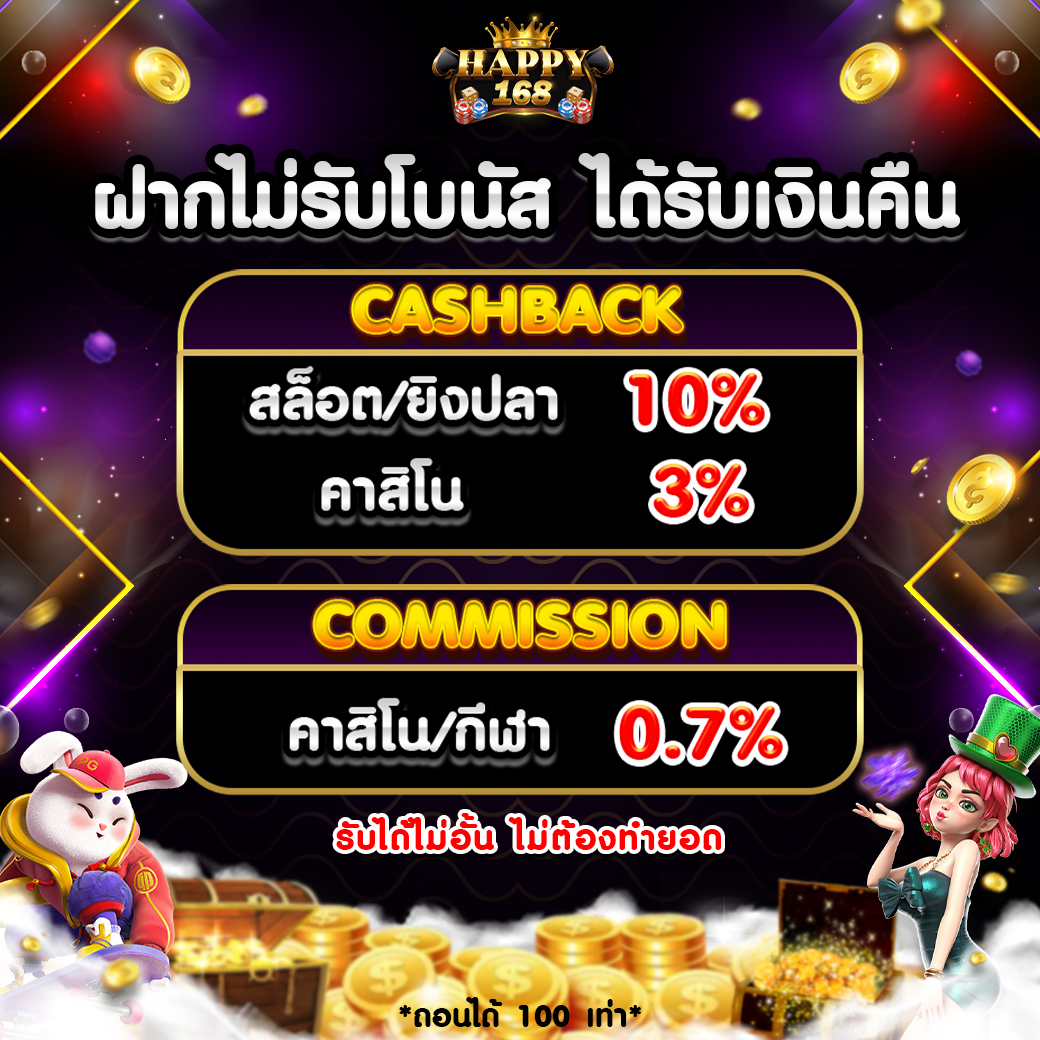
 Happy168 เว็บพนันสล็อตออนไลน์โดยเฉพาะอย่างยิ่ง โบนัสแตกง่าย โบนัสสูงสุด
Happy168 เว็บพนันสล็อตออนไลน์โดยเฉพาะอย่างยิ่ง โบนัสแตกง่าย โบนัสสูงสุด 1.รวมสล็อตจากหลายค่ายที่ได้รับความนิยมในที่เดียว เลือกเกมได้ไม่ซ้ำๆซากๆ
1.รวมสล็อตจากหลายค่ายที่ได้รับความนิยมในที่เดียว เลือกเกมได้ไม่ซ้ำๆซากๆ ยิ่งช่วงที่มี โปรฟรีสปิน หรือ เครดิตฟรี ผู้เล่นสามารถทดสอบเกมเหล่านี้ได้โดยแทบไม่ต้องลงทุนก่อน จึงเหมาะสมมากกับผู้ที่อยากหาเกมโบนัสเข้าไวๆ
ยิ่งช่วงที่มี โปรฟรีสปิน หรือ เครดิตฟรี ผู้เล่นสามารถทดสอบเกมเหล่านี้ได้โดยแทบไม่ต้องลงทุนก่อน จึงเหมาะสมมากกับผู้ที่อยากหาเกมโบนัสเข้าไวๆ นอกจากนั้นยังมีโปรคืนยอดเสีย รวมทั้งกิจกรรมสะสมยอดหมุน รับรางวัลเพิ่ม ซึ่งทั้งหมดทั้งปวงนี้วางแบบมาเพื่อ “คนเล่นสล็อต” โดยยิ่งไปกว่านั้น
นอกจากนั้นยังมีโปรคืนยอดเสีย รวมทั้งกิจกรรมสะสมยอดหมุน รับรางวัลเพิ่ม ซึ่งทั้งหมดทั้งปวงนี้วางแบบมาเพื่อ “คนเล่นสล็อต” โดยยิ่งไปกว่านั้น pussy888fun สบาย ไม่ต้องโหลดแอป เข้าเล่นผ่านหน้าเว็บไซต์ได้เลย รองรับทุกระบบ
pussy888fun สบาย ไม่ต้องโหลดแอป เข้าเล่นผ่านหน้าเว็บไซต์ได้เลย รองรับทุกระบบ


 เล่นมานานก็พลาดได้ จุดบกพร่องที่มือใหม่หรือมือเก๋ามักพลาดบน pgslot77
เล่นมานานก็พลาดได้ จุดบกพร่องที่มือใหม่หรือมือเก๋ามักพลาดบน pgslot77 ไม่ว่าคุณจะเล่นสล็อตกับ pg77 มานานเพียงใดก็ตาม จังหวะผิดพลาดก็เกิดขึ้นได้ สิ่งที่ผมอยากจะบอกมือใหม่ ทุกคนเลยก็คือ ข้อผิดพลาดนั้นไม่ใช่เรื่องร้ายแรงอะไรเลยครับผม คนที่เล่นมานานก็ยังพลาดได้เลย ซึ่งมันมิได้แสดงว่าเราเล่นไม่เก่ง แต่มันอาจจะเกิดขึ้นได้ก็เพราะความเข้าใจผิดหรือรู้เรื่องคลาดเคลื่อนด้วยก็ได้ พอไม่ถูกซ้ำๆนานๆเข้า มันก็เปลี่ยนเป็นความคุ้นเคยไปเลยนั่นเอง ในบทความนี้ ผมจะพาคุณมาดูเลยว่า ข้อผิดพลาดแบบไหนบ้างที่มือใหม่รวมทั้งมือเก๋ามักพลาดกัน
ไม่ว่าคุณจะเล่นสล็อตกับ pg77 มานานเพียงใดก็ตาม จังหวะผิดพลาดก็เกิดขึ้นได้ สิ่งที่ผมอยากจะบอกมือใหม่ ทุกคนเลยก็คือ ข้อผิดพลาดนั้นไม่ใช่เรื่องร้ายแรงอะไรเลยครับผม คนที่เล่นมานานก็ยังพลาดได้เลย ซึ่งมันมิได้แสดงว่าเราเล่นไม่เก่ง แต่มันอาจจะเกิดขึ้นได้ก็เพราะความเข้าใจผิดหรือรู้เรื่องคลาดเคลื่อนด้วยก็ได้ พอไม่ถูกซ้ำๆนานๆเข้า มันก็เปลี่ยนเป็นความคุ้นเคยไปเลยนั่นเอง ในบทความนี้ ผมจะพาคุณมาดูเลยว่า ข้อผิดพลาดแบบไหนบ้างที่มือใหม่รวมทั้งมือเก๋ามักพลาดกัน
 • เหมาะสมกับการเล่นเป็นตอนๆสั้น: เมื่อผมได้เล่นต่อเนื่อง ผมสังเกตว่า ฟีลเกมของแต่ละเกมเหมาะกับการเข้าไปเล่นเป็นบริเวณมากกว่าเล่นยาวๆต่อเนื่องกัน ถ้าเราอยากเล่นสัก 2 ชั่วโมง บางครั้งอาจจะแบ่งเป็น 4 รอบ รอบละ 30 นาทีก็ได้ครับ
• เหมาะสมกับการเล่นเป็นตอนๆสั้น: เมื่อผมได้เล่นต่อเนื่อง ผมสังเกตว่า ฟีลเกมของแต่ละเกมเหมาะกับการเข้าไปเล่นเป็นบริเวณมากกว่าเล่นยาวๆต่อเนื่องกัน ถ้าเราอยากเล่นสัก 2 ชั่วโมง บางครั้งอาจจะแบ่งเป็น 4 รอบ รอบละ 30 นาทีก็ได้ครับ
 เข้าใจค่า RTP ของเกม pgslotfish ได้มากขึ้น ไม่ได้บอกแค่จ่ายถี่ แต่บอกนิสัยเกมด้วย!
เข้าใจค่า RTP ของเกม pgslotfish ได้มากขึ้น ไม่ได้บอกแค่จ่ายถี่ แต่บอกนิสัยเกมด้วย! ค่า RTP ของเกม pgslot ควรดูยังไง สามารถใช้เลือกเกมได้จริงไหม?
ค่า RTP ของเกม pgslot ควรดูยังไง สามารถใช้เลือกเกมได้จริงไหม? เล่น pgfish แบบง่าย ๆ แค่เข้าใจค่า RTP ของเกมให้มากขึ้นก็พอ!
เล่น pgfish แบบง่าย ๆ แค่เข้าใจค่า RTP ของเกมให้มากขึ้นก็พอ!

 • ssru ทำกิจกรรมร่วมกับเพื่อนพ้องๆการที่น้องได้มีเพื่อนฝูงดีพร้อมช่วยเหลือเจือจุนกัน เป็นประเด็นสำคัญที่จะทำให้น้องจบการศึกษาไปยังภาคภูมิใจและก็มีความสุขในขณะที่เรียน ซึ่งกระบวนการทำกิจกรรมนั้น ไม่ว่าเป็นการเข้าชมรมต่างๆการเข้าค่าย เป็นต้น ทำให้จะได้โอกาสได้รู้จะเพื่อนใหม่ๆคณะอื่นอีกเยอะแยะ
• ssru ทำกิจกรรมร่วมกับเพื่อนพ้องๆการที่น้องได้มีเพื่อนฝูงดีพร้อมช่วยเหลือเจือจุนกัน เป็นประเด็นสำคัญที่จะทำให้น้องจบการศึกษาไปยังภาคภูมิใจและก็มีความสุขในขณะที่เรียน ซึ่งกระบวนการทำกิจกรรมนั้น ไม่ว่าเป็นการเข้าชมรมต่างๆการเข้าค่าย เป็นต้น ทำให้จะได้โอกาสได้รู้จะเพื่อนใหม่ๆคณะอื่นอีกเยอะแยะ

 สล็อตเครดิตฟรี ทำให้ผมไม่ต้องเสียเงินเสียทองลองผิดลองถูกเหมือนแต่ก่อน
สล็อตเครดิตฟรี ทำให้ผมไม่ต้องเสียเงินเสียทองลองผิดลองถูกเหมือนแต่ก่อน 2.กรอกข้อมูลรากฐาน ดังเช่นว่า เบอร์โทรศัพท์ ตั้งรหัสผ่าน และก็บัญชีธนาคารให้ถูก
2.กรอกข้อมูลรากฐาน ดังเช่นว่า เบอร์โทรศัพท์ ตั้งรหัสผ่าน และก็บัญชีธนาคารให้ถูก จังหวะรวมทั้งแบบการให้โบนัสไม่เหมือนกัน บางเกมเข้าโหมดฟรีสปินง่าย บางเกมเน้นย้ำจ่ายรางวัลถี่ การได้ทดลองแบบไม่จำเป็นต้องใช้เงินจริงช่วยให้ผมเลือกเกมที่เหมาะกับสไตล์การเล่นของตนเองเจริญขึ้น
จังหวะรวมทั้งแบบการให้โบนัสไม่เหมือนกัน บางเกมเข้าโหมดฟรีสปินง่าย บางเกมเน้นย้ำจ่ายรางวัลถี่ การได้ทดลองแบบไม่จำเป็นต้องใช้เงินจริงช่วยให้ผมเลือกเกมที่เหมาะกับสไตล์การเล่นของตนเองเจริญขึ้น ขอขอบคุณมากเว็ปไซต์
ขอขอบคุณมากเว็ปไซต์  pgslot เว็บตรงไม่ผ่านเอเย่นต์ และ เว็บเอเย่นต์ ต่างกันจริง ๆ ตรงไหน ถ้าต้องเลือก ควรเลือกแบบไหน
pgslot เว็บตรงไม่ผ่านเอเย่นต์ และ เว็บเอเย่นต์ ต่างกันจริง ๆ ตรงไหน ถ้าต้องเลือก ควรเลือกแบบไหน เวลาที่เรา จะเข้าเล่น เกมpgslot เรามักจะ สงสัย ในใจ ทันที ว่าควรเลือก เว็บแบบไหน เว็บตรงกับ เว็บเอเย่นต์มันต่างกันตรงไหน แล้วถ้าต้องตัดสินใจจริง ๆ จะเลือกยังไง สล็อต ดีล่ะ
เวลาที่เรา จะเข้าเล่น เกมpgslot เรามักจะ สงสัย ในใจ ทันที ว่าควรเลือก เว็บแบบไหน เว็บตรงกับ เว็บเอเย่นต์มันต่างกันตรงไหน แล้วถ้าต้องตัดสินใจจริง ๆ จะเลือกยังไง สล็อต ดีล่ะ แน่นอนว่าเว็บไซต์ ทั้ง 2 แบบ มีความคล้ายกัน มาก ๆ ระบบฝากถอนก็มีเหมือนกัน เกมก็ดูคล้าย ๆ กัน บางทีอาจจะเป็น เกมPG เหมือนกัน อีกด้วย แต่ความจริง ระบบหลังบ้าน ต่างกัน อย่างสิ้นเชิง เลยล่ะครับ fullslotpg และจุดต่าง ตรงนี้ กระทบ ต่อประสบการณ์ ในการเล่น ของเรา แบบชัด ๆ ถ้าอยากเห็นภาพ ก็มาลองกัน เลยครับ
แน่นอนว่าเว็บไซต์ ทั้ง 2 แบบ มีความคล้ายกัน มาก ๆ ระบบฝากถอนก็มีเหมือนกัน เกมก็ดูคล้าย ๆ กัน บางทีอาจจะเป็น เกมPG เหมือนกัน อีกด้วย แต่ความจริง ระบบหลังบ้าน ต่างกัน อย่างสิ้นเชิง เลยล่ะครับ fullslotpg และจุดต่าง ตรงนี้ กระทบ ต่อประสบการณ์ ในการเล่น ของเรา แบบชัด ๆ ถ้าอยากเห็นภาพ ก็มาลองกัน เลยครับ เว็บ bng55 เล่นแล้วเข้าใจ รีวิวจัดเต็ม เล่นเอง ของดีต้องการบอกต่อ
เว็บ bng55 เล่นแล้วเข้าใจ รีวิวจัดเต็ม เล่นเอง ของดีต้องการบอกต่อ ในช่วงเวลาที่ผมพบกับ สล็อต55 หนแรกไม่ได้มีสาเหตุมาจากการตั้งมั่นหาเว็บไซต์ใหม่ด้วย แต่ว่าเกิดขึ้นได้เนื่องมาจากระบบของเว็บเดิมที่ผมเล่นประจำกำเนิดล่มติดกันนับเป็นเวลาหลายวันจนหมดความทรหดอดทน พอลองเสิร์ชหาข้อเสนอในพันทิปและกลุ่มคาสิโนต่างๆก็มองเห็นชื่อ เว็บคาสิโนออนไลน์ เว็บไซต์คาสิโนออนไลน์ โผล่มาหลายคราวด้วยเสียงรีวิวในทางบวกเป็นส่วนมาก ในเวลานั้นก็ไม่ได้มุ่งมาดอะไรมากมาย สมัครเข้าไปดูเล่นๆเท่านั้น แต่ว่าพอใช้ทดลองใช้งานจริง กลายเป็นว่าเล่นติดยาวมาจนกระทั่งวันนี้แบบไม่ทันรู้ตัวเลย เพราะว่าเว็บไซต์มันใช้ง่าย ระบบลื่น โปรโมชั่นดูบริสุทธิ์ใจ ที่สำคัญเป็นเล่นแล้วไม่เคยรู้สึกว่าโดนเอาเปรียบเสมือนหลายเว็บก่อนหน้านี้
ในช่วงเวลาที่ผมพบกับ สล็อต55 หนแรกไม่ได้มีสาเหตุมาจากการตั้งมั่นหาเว็บไซต์ใหม่ด้วย แต่ว่าเกิดขึ้นได้เนื่องมาจากระบบของเว็บเดิมที่ผมเล่นประจำกำเนิดล่มติดกันนับเป็นเวลาหลายวันจนหมดความทรหดอดทน พอลองเสิร์ชหาข้อเสนอในพันทิปและกลุ่มคาสิโนต่างๆก็มองเห็นชื่อ เว็บคาสิโนออนไลน์ เว็บไซต์คาสิโนออนไลน์ โผล่มาหลายคราวด้วยเสียงรีวิวในทางบวกเป็นส่วนมาก ในเวลานั้นก็ไม่ได้มุ่งมาดอะไรมากมาย สมัครเข้าไปดูเล่นๆเท่านั้น แต่ว่าพอใช้ทดลองใช้งานจริง กลายเป็นว่าเล่นติดยาวมาจนกระทั่งวันนี้แบบไม่ทันรู้ตัวเลย เพราะว่าเว็บไซต์มันใช้ง่าย ระบบลื่น โปรโมชั่นดูบริสุทธิ์ใจ ที่สำคัญเป็นเล่นแล้วไม่เคยรู้สึกว่าโดนเอาเปรียบเสมือนหลายเว็บก่อนหน้านี้ bng55 เว็บที่ใบหน้าเรียบง่ายแม้กระนั้นระบบด้านในกลับลื่นกว่าเว็บที่ตกแต่งหรูหรา
bng55 เว็บที่ใบหน้าเรียบง่ายแม้กระนั้นระบบด้านในกลับลื่นกว่าเว็บที่ตกแต่งหรูหรา
 เว็บไซต์สล็อต bonanza99 ที่ใครก็เอ่ยถึง ทดสอบเล่นฟรี แถมได้เล่นเกมใหม่ก่อนคนไหนกันแน่จริง
เว็บไซต์สล็อต bonanza99 ที่ใครก็เอ่ยถึง ทดสอบเล่นฟรี แถมได้เล่นเกมใหม่ก่อนคนไหนกันแน่จริง มีเกมอะไรให้เล่นใน bonanza99 บ้าง ค่ายมากจนถึงเล่นไม่ครบในวันเดียว
มีเกมอะไรให้เล่นใน bonanza99 บ้าง ค่ายมากจนถึงเล่นไม่ครบในวันเดียว รีวิวเว็บไซต์สล็อต jinda55 ที่แจกหนักที่สุดในตอนต้นปี 2026 พร้อมโปรโมชั่นสุดเร่าร้อน !
รีวิวเว็บไซต์สล็อต jinda55 ที่แจกหนักที่สุดในตอนต้นปี 2026 พร้อมโปรโมชั่นสุดเร่าร้อน ! สิ่งที่ผมต้องใจที่สุดไม่ใช่แค่การแตกของเกม แต่คือความเร็วสำหรับเพื่อการถอนเงิน ผมกดถอนตอนสองนาฬิกา และเงินเข้าบัญชีภายใน 50 วินาทีโดยไม่มีแอดมินทักถามหรือขอข้อมูลอะไรเพิ่มอีกเลย
สิ่งที่ผมต้องใจที่สุดไม่ใช่แค่การแตกของเกม แต่คือความเร็วสำหรับเพื่อการถอนเงิน ผมกดถอนตอนสองนาฬิกา และเงินเข้าบัญชีภายใน 50 วินาทีโดยไม่มีแอดมินทักถามหรือขอข้อมูลอะไรเพิ่มอีกเลย 3. ถ้าเล่นเสีย จะได้รับอะไรกลับมาหรือไม่
3. ถ้าเล่นเสีย จะได้รับอะไรกลับมาหรือไม่
 918kiss ยอดเยี่ยมในแอปสล็อตที่ผมใช้งานสม่ำเสมอช้านานที่สุด และในปีนี้ผมได้มีโอกาส ดาวน์โหลด 918kiss เวอร์ชั่นปัจจุบัน 2026 มาใช้งานจริง ซึ่งบอกตรงๆว่าประสบการณ์ดียิ่งกว่ารุ่นก่อนพอควร อีกทั้งในเรื่องความมีประสิทธิภาพ ความเร็ว รวมทั้งฟีพบร์ใหม่ๆที่ตอบโจทย์ผู้เล่นมากขึ้น
918kiss ยอดเยี่ยมในแอปสล็อตที่ผมใช้งานสม่ำเสมอช้านานที่สุด และในปีนี้ผมได้มีโอกาส ดาวน์โหลด 918kiss เวอร์ชั่นปัจจุบัน 2026 มาใช้งานจริง ซึ่งบอกตรงๆว่าประสบการณ์ดียิ่งกว่ารุ่นก่อนพอควร อีกทั้งในเรื่องความมีประสิทธิภาพ ความเร็ว รวมทั้งฟีพบร์ใหม่ๆที่ตอบโจทย์ผู้เล่นมากขึ้น allbet24hr เกมให้เลือกเล่นมากกว่า 2,000 เกม ถือเป็นจุดแปลงสำคัญมาก เพราะเหตุว่ามันไม่ใช่แค่ “เยอะแยะ” แม้กระนั้นเป็น “ครบ” รวมทั้ง “ตอบปัญหา” ผู้เล่นทุกสไตล์จริงๆช่วงแรกผมเป็นคนเล่นสล็อตแบบเลือกเกมเดิมๆด้วยเหตุว่ากลัวว่าเกมใหม่จะเล่นยาก
allbet24hr เกมให้เลือกเล่นมากกว่า 2,000 เกม ถือเป็นจุดแปลงสำคัญมาก เพราะเหตุว่ามันไม่ใช่แค่ “เยอะแยะ” แม้กระนั้นเป็น “ครบ” รวมทั้ง “ตอบปัญหา” ผู้เล่นทุกสไตล์จริงๆช่วงแรกผมเป็นคนเล่นสล็อตแบบเลือกเกมเดิมๆด้วยเหตุว่ากลัวว่าเกมใหม่จะเล่นยาก

 funny18 เว็บไซต์สล็อตที่คนเล่นจริงต่างยกนิ้วให้เรื่องแตกง่าย เบิกเงินได้ไว และบริการแบบมือโปร
funny18 เว็บไซต์สล็อตที่คนเล่นจริงต่างยกนิ้วให้เรื่องแตกง่าย เบิกเงินได้ไว และบริการแบบมือโปร ถ้าหากจะให้เล่าความรู้สึกที่มีต่อการเล่นสล็อตในพักหลัง ต้องยอมรับตรงๆว่าหลายเว็บที่เคยเล่นfunny18 ให้ประสบการณ์ค่อนข้างห่วย ไม่ว่าจะเป็นการล็อกยูส ระบบถอนไม่เข้า โปรโมชั่นที่มีเงื่อนไขเยอะเกินไป หรือแอดมินตอบช้าแบบปลดปล่อยให้คอยเกือบจะชั่วโมง แต่ว่าความรู้สึกพวกนั้นก็เปลี่ยนไปทันทีภายหลังได้ลองเล่นกับ PG
ถ้าหากจะให้เล่าความรู้สึกที่มีต่อการเล่นสล็อตในพักหลัง ต้องยอมรับตรงๆว่าหลายเว็บที่เคยเล่นfunny18 ให้ประสบการณ์ค่อนข้างห่วย ไม่ว่าจะเป็นการล็อกยูส ระบบถอนไม่เข้า โปรโมชั่นที่มีเงื่อนไขเยอะเกินไป หรือแอดมินตอบช้าแบบปลดปล่อยให้คอยเกือบจะชั่วโมง แต่ว่าความรู้สึกพวกนั้นก็เปลี่ยนไปทันทีภายหลังได้ลองเล่นกับ PG

 เพราะอะไรจะต้องเล่นสล็อตออนไลน์กับ 918kiss เว็บไซต์ตรง
เพราะอะไรจะต้องเล่นสล็อตออนไลน์กับ 918kiss เว็บไซต์ตรง • ระบบโบนัสแน่ชัด: ฟรีสปินเข้าไม่ยาก ไม่ต้องรอคอยรอบลึกก็เข้าได้ แถมบางเกม918kiss ยังมีตัวคูณดำเนินงานร่วมกับฟรีสปิน ทำให้ท่านมีโอกาสรับกำไรเยอะขึ้นเรื่อยๆหลายเท่า บางเกมมีโบนัสซ้อนมอีก ผมบอกเลยว่า ได้หลายต่อจากสปินเดียวได้แน่นอน
• ระบบโบนัสแน่ชัด: ฟรีสปินเข้าไม่ยาก ไม่ต้องรอคอยรอบลึกก็เข้าได้ แถมบางเกม918kiss ยังมีตัวคูณดำเนินงานร่วมกับฟรีสปิน ทำให้ท่านมีโอกาสรับกำไรเยอะขึ้นเรื่อยๆหลายเท่า บางเกมมีโบนัสซ้อนมอีก ผมบอกเลยว่า ได้หลายต่อจากสปินเดียวได้แน่นอน ผู้คนจำนวนมากเรียก 918kissauto ว่าเป็นปรมาจารย์ด้านสล็อตมือถือแบบสุดๆเนื่องจากพวกเราคือค่ายเกมแรกๆที่พัฒนารวมทั้งดีไซน์เกมสล็อตสำหรับโทรศัพท์มือถือโดยเฉพาะ เนื่องจากว่าเราเชื่อกันว่า โทรศัพท์มือถือจะเป็นเครื่องใช้ไม้สอยหลักที่ทุกคนจะใช้เล่นสล็อตนั่นเอง
ผู้คนจำนวนมากเรียก 918kissauto ว่าเป็นปรมาจารย์ด้านสล็อตมือถือแบบสุดๆเนื่องจากพวกเราคือค่ายเกมแรกๆที่พัฒนารวมทั้งดีไซน์เกมสล็อตสำหรับโทรศัพท์มือถือโดยเฉพาะ เนื่องจากว่าเราเชื่อกันว่า โทรศัพท์มือถือจะเป็นเครื่องใช้ไม้สอยหลักที่ทุกคนจะใช้เล่นสล็อตนั่นเอง สูตรเล่นสล็อตแบบมือโปร 918kissauto Kissat.app 7 ก.พ. 2026 Anastasia คาสิโนออนไลน์ 918kissauto มาแรงที่สุด Top 70
สูตรเล่นสล็อตแบบมือโปร 918kissauto Kissat.app 7 ก.พ. 2026 Anastasia คาสิโนออนไลน์ 918kissauto มาแรงที่สุด Top 70
 ก่อนหน้านี้ผมเป็นผู้ที่แทงหวยกับเจ้ามือแถวบ้านเป็นหลัก แทงหวยออนไลน์ ไม่ว่าจะหวยรัฐบาลหรือสลากกินแบ่งลาวก็จะจดโพย ส่งไลน์ โอนเงินแบบงงงันๆบางคราวก็ลืมเลขที่แทง บางคราวเจ้ามือก็จ่ายช้า หรือห่วยกว่านั้นเป็นงวดที่ถูก ดันกล่าวว่ายังไม่เปิดรับงวดนั้น ท้ายที่สุดเลยเริ่มมองหาเว็บไซต์หวยออนไลน์ที่ระบบกระจ่างและก็เชื่อใจได้จริงจัง
ก่อนหน้านี้ผมเป็นผู้ที่แทงหวยกับเจ้ามือแถวบ้านเป็นหลัก แทงหวยออนไลน์ ไม่ว่าจะหวยรัฐบาลหรือสลากกินแบ่งลาวก็จะจดโพย ส่งไลน์ โอนเงินแบบงงงันๆบางคราวก็ลืมเลขที่แทง บางคราวเจ้ามือก็จ่ายช้า หรือห่วยกว่านั้นเป็นงวดที่ถูก ดันกล่าวว่ายังไม่เปิดรับงวดนั้น ท้ายที่สุดเลยเริ่มมองหาเว็บไซต์หวยออนไลน์ที่ระบบกระจ่างและก็เชื่อใจได้จริงจัง หากคุณเป็นคนนึงที่เคยผิดหวังกับเว็บไซต์หวยที่เคยเล่นมา หรือกำลังมองหาเว็บไซต์ที่มั่นอาจและจ่ายจริงแบบไม่ต้องลุ้นว่าจะโดนเท ผมกล้าพูดเลยว่า เว็บซื้อหวย เป็นเว็บไซต์ที่คุณควรทดลองเปิดใจเล่นมองสักครั้ง แล้วคุณอาจจะไม่ต้องการที่จะอยากกลับไปเขียนโพยแบบเดิมอีกเลย
หากคุณเป็นคนนึงที่เคยผิดหวังกับเว็บไซต์หวยที่เคยเล่นมา หรือกำลังมองหาเว็บไซต์ที่มั่นอาจและจ่ายจริงแบบไม่ต้องลุ้นว่าจะโดนเท ผมกล้าพูดเลยว่า เว็บซื้อหวย เป็นเว็บไซต์ที่คุณควรทดลองเปิดใจเล่นมองสักครั้ง แล้วคุณอาจจะไม่ต้องการที่จะอยากกลับไปเขียนโพยแบบเดิมอีกเลย สล็อตเว็บใหญ่ เว็บซื้อหวย 24 NOV 25 Lawrence casino lotto77 แตกหนัก Top 58
สล็อตเว็บใหญ่ เว็บซื้อหวย 24 NOV 25 Lawrence casino lotto77 แตกหนัก Top 58 เมื่อพูดถึงคาสิโนออนไลน์ คนไม่ใช่น้อยมักจะมีความคิดว่าเป็นเรื่องซับซ้อน เข้าถึงยาก จะต้องมีความรู้ หรือใช้เงินมากมายถึงจะเล่นได้ ซึ่งความนึกคิดกลุ่มนี้อาจเคยจริงในอดีตกาล แม้กระนั้นไม่ใช่กับเว็บไซต์
เมื่อพูดถึงคาสิโนออนไลน์ คนไม่ใช่น้อยมักจะมีความคิดว่าเป็นเรื่องซับซ้อน เข้าถึงยาก จะต้องมีความรู้ หรือใช้เงินมากมายถึงจะเล่นได้ ซึ่งความนึกคิดกลุ่มนี้อาจเคยจริงในอดีตกาล แม้กระนั้นไม่ใช่กับเว็บไซต์ 


 เกม พุซซี่888 เล่นบ่อยมากแต่ว่าไม่ค่อยแตก เป็นเพราะเหตุใดกันแน่?
เกม พุซซี่888 เล่นบ่อยมากแต่ว่าไม่ค่อยแตก เป็นเพราะเหตุใดกันแน่? 2. เหตุเชิงเกมและเทคนิค
2. เหตุเชิงเกมและเทคนิค

 การเล่นคาสิโนออนไลน์ไม่ใช่เรื่องใหม่สำหรับคนไม่ใช่น้อย แม้กระนั้นสำหรับผม การได้สัมผัส เกมคาสิโนออนไลน์ยอดฮิตชั้น 1 macau69 ถือเป็นประสบการณ์ที่ตื่นเต้นแล้วก็เบิกบานมากยิ่งกว่าที่คิด ทุกครั้งที่ผมเข้าสู่แพลตฟอร์มเกม ไม่ว่าจะเป็นบนโทรศัพท์เคลื่อนที่หรือคอมพิวเตอร์ ผมรู้สึกราวกับกำลังเดินเข้าไปยังคาสิโนสุดหรูในลาสเวกัส แต่ทั้งปวงอยู่เบื้องหน้าหน้าจอของผมเอง
การเล่นคาสิโนออนไลน์ไม่ใช่เรื่องใหม่สำหรับคนไม่ใช่น้อย แม้กระนั้นสำหรับผม การได้สัมผัส เกมคาสิโนออนไลน์ยอดฮิตชั้น 1 macau69 ถือเป็นประสบการณ์ที่ตื่นเต้นแล้วก็เบิกบานมากยิ่งกว่าที่คิด ทุกครั้งที่ผมเข้าสู่แพลตฟอร์มเกม ไม่ว่าจะเป็นบนโทรศัพท์เคลื่อนที่หรือคอมพิวเตอร์ ผมรู้สึกราวกับกำลังเดินเข้าไปยังคาสิโนสุดหรูในลาสเวกัส แต่ทั้งปวงอยู่เบื้องหน้าหน้าจอของผมเอง macau69 สมัคร ขั้นตอนไม่สลับซับซ้อน สนุกสนานทุกสปิน เริ่ม 1 บาทก็แตกได้
macau69 สมัคร ขั้นตอนไม่สลับซับซ้อน สนุกสนานทุกสปิน เริ่ม 1 บาทก็แตกได้
 เว็บสล็อต เครดิตฟรี ไม่ต้องฝาก สนุกได้ไม่น่าเบื่อ! ทดสอบเล่นฟรีไม่เสียเงินก่อนเริ่มพนันจริง
เว็บสล็อต เครดิตฟรี ไม่ต้องฝาก สนุกได้ไม่น่าเบื่อ! ทดสอบเล่นฟรีไม่เสียเงินก่อนเริ่มพนันจริง


 ทุกโปรโมชั่นสามารถกดรับเองได้จากหน้าเว็บไซต์ รวมทั้งถอนเงินได้จริงตามข้อตกลง โดยเฉพาะอย่างยิ่งโบนัส vvip1668 เครดิตฟรี ที่ได้รับความนิยมสูงสุดเพราะเหตุว่าไม่ต้องฝากก่อนและไม่จำเป็นต้องทำเทิร์นมาก.
ทุกโปรโมชั่นสามารถกดรับเองได้จากหน้าเว็บไซต์ รวมทั้งถอนเงินได้จริงตามข้อตกลง โดยเฉพาะอย่างยิ่งโบนัส vvip1668 เครดิตฟรี ที่ได้รับความนิยมสูงสุดเพราะเหตุว่าไม่ต้องฝากก่อนและไม่จำเป็นต้องทำเทิร์นมาก.
 shinobi24 คือหนึ่งในเว็บไซต์สล็อตออนไลน์ที่กำลังเป็นที่นิยมสูงสุดในตอนนี้ ด้วยจุดเด่นเรื่องระบบที่นำสมัย ปลอดภัย รวมทั้งใช้งานง่าย เหมาะกับผู้เล่นทุกเพศทุกวัย ไม่ว่าจะเป็นมือใหม่หรือมือโปรก็สามารถเข้ามาร่วมสนุกสนานได้อย่างไร้ขีดจำกัด การ shinobi24 สมัคร ทำเป็นรวดเร็วทันใจข้างในไม่กี่ขั้นตอน พร้อมรับโบนัสต้อนรับแล้วก็ shinobi24 เครดิตฟรี ในทันทีหลังลงทะเบียนสมัครสมาชิก เพื่อผู้เล่นสามารถเริ่มต้นการเดิมพันได้อย่างแน่ใจตั้งแต่คราวแรก
shinobi24 คือหนึ่งในเว็บไซต์สล็อตออนไลน์ที่กำลังเป็นที่นิยมสูงสุดในตอนนี้ ด้วยจุดเด่นเรื่องระบบที่นำสมัย ปลอดภัย รวมทั้งใช้งานง่าย เหมาะกับผู้เล่นทุกเพศทุกวัย ไม่ว่าจะเป็นมือใหม่หรือมือโปรก็สามารถเข้ามาร่วมสนุกสนานได้อย่างไร้ขีดจำกัด การ shinobi24 สมัคร ทำเป็นรวดเร็วทันใจข้างในไม่กี่ขั้นตอน พร้อมรับโบนัสต้อนรับแล้วก็ shinobi24 เครดิตฟรี ในทันทีหลังลงทะเบียนสมัครสมาชิก เพื่อผู้เล่นสามารถเริ่มต้นการเดิมพันได้อย่างแน่ใจตั้งแต่คราวแรก ระบบ shinobi24 สมัคร ถูกออกแบบมาให้ใช้งานง่าย ไม่ซับซ้อน สามารถสมัครผ่านหน้าเว็บไซต์โดยตรง กรอกข้อมูลเพียงไม่กี่ช่องแล้วก็รับรองเบอร์โทรศัพท์ ก็สามารถ shinobi24 เข้าสู่ระบบ ได้ในทันที โดยไม่ต้องผ่านแอดไม่น เพิ่มความสะดวกให้กับผู้เล่นทุกคน รองรับอีกทั้งบนโทรศัพท์มือถือและคอมพิวเตอร์ เพื่อการเข้าถึงที่สบายทุกเมื่อ
ระบบ shinobi24 สมัคร ถูกออกแบบมาให้ใช้งานง่าย ไม่ซับซ้อน สามารถสมัครผ่านหน้าเว็บไซต์โดยตรง กรอกข้อมูลเพียงไม่กี่ช่องแล้วก็รับรองเบอร์โทรศัพท์ ก็สามารถ shinobi24 เข้าสู่ระบบ ได้ในทันที โดยไม่ต้องผ่านแอดไม่น เพิ่มความสะดวกให้กับผู้เล่นทุกคน รองรับอีกทั้งบนโทรศัพท์มือถือและคอมพิวเตอร์ เพื่อการเข้าถึงที่สบายทุกเมื่อ
 หากแม้เกมสล็อตจะเป็นเกมที่อาศัยดวง แต่ถ้าเกิดผู้เล่นมีวิธีการการเล่นที่ดี ช่องทางสำหรับในการทำเงินก็จะสูงขึ้นหลายเท่า ซึ่งใน autoslot168 มีระบบระเบียบทดลองเล่นฟรีที่เปิดให้ผู้เล่นสามารถใช้ autoslot168 เครดิตฟรี เพื่อฝึกฝนก่อนลงสนามจริงได้
หากแม้เกมสล็อตจะเป็นเกมที่อาศัยดวง แต่ถ้าเกิดผู้เล่นมีวิธีการการเล่นที่ดี ช่องทางสำหรับในการทำเงินก็จะสูงขึ้นหลายเท่า ซึ่งใน autoslot168 มีระบบระเบียบทดลองเล่นฟรีที่เปิดให้ผู้เล่นสามารถใช้ autoslot168 เครดิตฟรี เพื่อฝึกฝนก่อนลงสนามจริงได้ autoslot168 เป็นเว็บตรงไม่ผ่านเอเย่นต์ที่ได้รับใบอนุมัติจากหน่วยงานเกมออนไลน์ระดับสากล มีระบบระเบียบรักษาความปลอดภัยระดับสูงรวมทั้งทีมงานสำรวจกระบวนการทำธุรกรรมตลอด 1 วัน ทำให้ผู้เล่นมั่นใจได้ว่าทุกการเดิมพันจะโปร่งใส ปลอดภัย และก็สามารถเบิกเงินได้จริงทุกยอด
autoslot168 เป็นเว็บตรงไม่ผ่านเอเย่นต์ที่ได้รับใบอนุมัติจากหน่วยงานเกมออนไลน์ระดับสากล มีระบบระเบียบรักษาความปลอดภัยระดับสูงรวมทั้งทีมงานสำรวจกระบวนการทำธุรกรรมตลอด 1 วัน ทำให้ผู้เล่นมั่นใจได้ว่าทุกการเดิมพันจะโปร่งใส ปลอดภัย และก็สามารถเบิกเงินได้จริงทุกยอด แนวทางการ สมัคร betwin666 ทำเป็นง่ายแล้วก็รวดเร็วทันใจ ผู้เล่นเพียงแต่กรอกข้อมูลไม่กี่ช่อง อาทิเช่น เบอร์โทรศัพท์ บัญชีธนาคาร แล้วก็ตั้งรหัสผ่าน หลังจากนั้นการันตี OTP ก็สามารถเริ่มเล่นได้ในทันที ไม่ต้องผ่านแอดไม่น และไม่ต้องคอยอนุมัติ ระบบถูกออกแบบให้ไม่เป็นอันตราย 100% ด้วยเทคโนโลยีเข้ารหัสข้อมูล SSL เชื่อมั่นได้ว่าข้อมูลส่วนตัวจะไม่รั่วไหล
แนวทางการ สมัคร betwin666 ทำเป็นง่ายแล้วก็รวดเร็วทันใจ ผู้เล่นเพียงแต่กรอกข้อมูลไม่กี่ช่อง อาทิเช่น เบอร์โทรศัพท์ บัญชีธนาคาร แล้วก็ตั้งรหัสผ่าน หลังจากนั้นการันตี OTP ก็สามารถเริ่มเล่นได้ในทันที ไม่ต้องผ่านแอดไม่น และไม่ต้องคอยอนุมัติ ระบบถูกออกแบบให้ไม่เป็นอันตราย 100% ด้วยเทคโนโลยีเข้ารหัสข้อมูล SSL เชื่อมั่นได้ว่าข้อมูลส่วนตัวจะไม่รั่วไหล ในยุคที่คาสิโนออนไลน์มีมากมายก่ายกอง การเลือกเว็บไซต์ที่ปลอดภัยและคุ้มถือเป็นสิ่งจำเป็น niseko168
ในยุคที่คาสิโนออนไลน์มีมากมายก่ายกอง การเลือกเว็บไซต์ที่ปลอดภัยและคุ้มถือเป็นสิ่งจำเป็น niseko168  ความพิเศษของระบบเข้าระบบ
ความพิเศษของระบบเข้าระบบ สรุปภาพรวมของ niseko168
สรุปภาพรวมของ niseko168
 ถ้าคุณกำลังมองหาแหล่งรวมเกมสล็อตและก็คาสิโนออนไลน์ที่ใช้งานง่ายแล้วก็มีโปรโมชั่นรองรับผู้เล่นใหม่ ชื่อ ambfine88 มักเป็นคำค้นที่ได้รับความนิยม เนื้อหานี้วางแบบมาเพื่อข้อมูลครบสมบูรณ์ตั้งแต่ภาพรวม ฟีพบร์หลัก แนวทาง ambfine88 สมัคร แนวทางการไม่มีอันตรายสำหรับ ambfine88 เข้าสู่ระบบ การใช้สิทธิประโยชน์ดังเช่น
ถ้าคุณกำลังมองหาแหล่งรวมเกมสล็อตและก็คาสิโนออนไลน์ที่ใช้งานง่ายแล้วก็มีโปรโมชั่นรองรับผู้เล่นใหม่ ชื่อ ambfine88 มักเป็นคำค้นที่ได้รับความนิยม เนื้อหานี้วางแบบมาเพื่อข้อมูลครบสมบูรณ์ตั้งแต่ภาพรวม ฟีพบร์หลัก แนวทาง ambfine88 สมัคร แนวทางการไม่มีอันตรายสำหรับ ambfine88 เข้าสู่ระบบ การใช้สิทธิประโยชน์ดังเช่น  ambfine88 = แพลตฟอร์มเกมออนไลน์ที่ให้บริการสล็อตแล้วก็คาสิโนสด; เริ่มจากสมัคร (ambfine88 สมัคร) การันตีตัวตน แล้วล็อกอิน (ambfine88 เข้าระบบ); ดูโปรโมชั่นอย่าง ambfine88 เครดิตฟรี แต่จำต้องอ่านข้อแม้ก่อนรับ; ทดสอบระบบด้วยเครดิตฟรีหรือฝากนิดหน่อยก่อนลงทุนมาก
ambfine88 = แพลตฟอร์มเกมออนไลน์ที่ให้บริการสล็อตแล้วก็คาสิโนสด; เริ่มจากสมัคร (ambfine88 สมัคร) การันตีตัวตน แล้วล็อกอิน (ambfine88 เข้าระบบ); ดูโปรโมชั่นอย่าง ambfine88 เครดิตฟรี แต่จำต้องอ่านข้อแม้ก่อนรับ; ทดสอบระบบด้วยเครดิตฟรีหรือฝากนิดหน่อยก่อนลงทุนมาก อ่านรีวิวจากหลายแหล่ง ทั้งฟอรัม กรุ๊ปโซเชียล รวมทั้งเว็บไซต์รีวิวที่เป็นกลาง
อ่านรีวิวจากหลายแหล่ง ทั้งฟอรัม กรุ๊ปโซเชียล รวมทั้งเว็บไซต์รีวิวที่เป็นกลาง
 hengheng989 เป็นเว็บพนันออนไลน์ที่กำลังเป็นที่นิยมอย่างยิ่งในประเทศไทย ด้วยระบบการให้บริการที่ครบวงจรและก็ไม่เป็นอันตรายที่สุด เปิดให้ผู้เล่นสามารถ hengheng989 สมัคร ได้ฟรีข้างในไม่กี่วินาที ผ่านระบบออโต้ที่รวดเร็ว รวมทั้งยังมีระบบ hengheng989 เข้าระบบ ที่เสถียร รองรับทั้งยังโทรศัพท์เคลื่อนที่รวมทั้งคอมพิวเตอร์ พร้อมโปรโมชั่นสุดพิเศษอย่าง hengheng989 เครดิตฟรี สำหรับสมาชิกใหม่และเก่า เพื่อคุณสนุกกับการพนันได้อย่างคุ้มค่าในวันแล้ววันเล่า
hengheng989 เป็นเว็บพนันออนไลน์ที่กำลังเป็นที่นิยมอย่างยิ่งในประเทศไทย ด้วยระบบการให้บริการที่ครบวงจรและก็ไม่เป็นอันตรายที่สุด เปิดให้ผู้เล่นสามารถ hengheng989 สมัคร ได้ฟรีข้างในไม่กี่วินาที ผ่านระบบออโต้ที่รวดเร็ว รวมทั้งยังมีระบบ hengheng989 เข้าระบบ ที่เสถียร รองรับทั้งยังโทรศัพท์เคลื่อนที่รวมทั้งคอมพิวเตอร์ พร้อมโปรโมชั่นสุดพิเศษอย่าง hengheng989 เครดิตฟรี สำหรับสมาชิกใหม่และเก่า เพื่อคุณสนุกกับการพนันได้อย่างคุ้มค่าในวันแล้ววันเล่า
 infyplus88 เป็นเว็บไซต์ตรงไม่ผ่านเอเย่นต์ที่ได้รับการพัฒนาให้ตอบโจทย์ผู้เล่นสมัยใหม่ ด้วยเทคโนโลยีที่เร็วทันใจ ไม่เป็นอันตราย รวมทั้งเข้าถึงง่าย ไม่ว่าจะเป็นเกมสล็อต คาสิโนสด หรือเกมเดิมพันแบบอย่างอื่นๆก็มีครบในที่เดียว พร้อมระบบฝากถอนอัตโนมัติที่รวดเร็วและโปร่งใส
infyplus88 เป็นเว็บไซต์ตรงไม่ผ่านเอเย่นต์ที่ได้รับการพัฒนาให้ตอบโจทย์ผู้เล่นสมัยใหม่ ด้วยเทคโนโลยีที่เร็วทันใจ ไม่เป็นอันตราย รวมทั้งเข้าถึงง่าย ไม่ว่าจะเป็นเกมสล็อต คาสิโนสด หรือเกมเดิมพันแบบอย่างอื่นๆก็มีครบในที่เดียว พร้อมระบบฝากถอนอัตโนมัติที่รวดเร็วและโปร่งใส
 เทคนิคที่ควรรู้ก่อนเริ่มพนัน
เทคนิคที่ควรรู้ก่อนเริ่มพนัน บทสรุป:
บทสรุป:  ผลสรุป: zaf888 เว็บไซต์ตรงประสิทธิภาพที่ตอบปัญหานักพนันยุคสมัยใหม่
ผลสรุป: zaf888 เว็บไซต์ตรงประสิทธิภาพที่ตอบปัญหานักพนันยุคสมัยใหม่ สิ่งที่ทำให้ gowingo เด่นและก็เป็นที่นิยมในกลุ่มนักปั่นสล็อต เป็นการพัฒนาเว็บให้ตอบโจทย์ผู้เล่นทุกประเภท ทั้งด้านระบบ ความปลอดภัย รวมทั้งความคุ้มค่าของโปรโมชั่น ซึ่งทำให้เว็บแห่งนี้ขึ้นแท่นเว็บสล็อตลำดับต้นๆของเอเชีย
สิ่งที่ทำให้ gowingo เด่นและก็เป็นที่นิยมในกลุ่มนักปั่นสล็อต เป็นการพัฒนาเว็บให้ตอบโจทย์ผู้เล่นทุกประเภท ทั้งด้านระบบ ความปลอดภัย รวมทั้งความคุ้มค่าของโปรโมชั่น ซึ่งทำให้เว็บแห่งนี้ขึ้นแท่นเว็บสล็อตลำดับต้นๆของเอเชีย ข้อดีของระบบเข้าระบบ 928COINS
ข้อดีของระบบเข้าระบบ 928COINS pglucky เป็นเว็บสล็อตออนไลน์ที่ตอบปัญหาผู้เล่นยุคสมัยใหม่ก้าวหน้าที่สุด ด้วยระบบ pglucky สมัคร ที่ใช้งานง่าย ระบบ pglucky เข้าสู่ระบบ ที่เสถียร และก็โปรโมชั่น pglucky เครดิตฟรี ที่แจกจริงวันแล้ววันเล่า มาพร้อมเกมประสิทธิภาพจากค่าย PG SLOT ที่ทั้งยังสนุกและก็ทำเงินได้จริง
pglucky เป็นเว็บสล็อตออนไลน์ที่ตอบปัญหาผู้เล่นยุคสมัยใหม่ก้าวหน้าที่สุด ด้วยระบบ pglucky สมัคร ที่ใช้งานง่าย ระบบ pglucky เข้าสู่ระบบ ที่เสถียร และก็โปรโมชั่น pglucky เครดิตฟรี ที่แจกจริงวันแล้ววันเล่า มาพร้อมเกมประสิทธิภาพจากค่าย PG SLOT ที่ทั้งยังสนุกและก็ทำเงินได้จริง
 สล็อต168 เว็บตรงไม่ผ่านเอเย่นต์เดียวในไทย เอนจอยกับการทำเงินแบบจัดเต็ม ฝากถอนไม่มีอย่างน้อย ทดลองเลย!
สล็อต168 เว็บตรงไม่ผ่านเอเย่นต์เดียวในไทย เอนจอยกับการทำเงินแบบจัดเต็ม ฝากถอนไม่มีอย่างน้อย ทดลองเลย! ความชื่นชอบของเกม สล็อต168 ที่คุณอาจจะไม่รู้ มาทดลองดูกันเลย!
ความชื่นชอบของเกม สล็อต168 ที่คุณอาจจะไม่รู้ มาทดลองดูกันเลย! • ความสะดวกสบาย: เกม สล็อต168 ในปัจจุบัน จะได้รับความนิยมอย่างมากในการเล่นบนเครื่องใช้ไม้สอยอย่างโทรศัพท์มือถือ เพราะเหตุว่ามันสามารถเล่นที่แห่งไหนก็ได้ เล่นตอนไหนก็ได้ ไม่ว่าคุณจะเดินทาง ทานข้าว หรือกำลังเข้านอน ก็สามารถเปิดโทรศัพท์มือถือรวมทั้งเล่นสล็อตออนไลน์ได้แบบชิวๆสบายต่อการใช้แรงงานแบบสุดๆเลยล่ะขอรับ
• ความสะดวกสบาย: เกม สล็อต168 ในปัจจุบัน จะได้รับความนิยมอย่างมากในการเล่นบนเครื่องใช้ไม้สอยอย่างโทรศัพท์มือถือ เพราะเหตุว่ามันสามารถเล่นที่แห่งไหนก็ได้ เล่นตอนไหนก็ได้ ไม่ว่าคุณจะเดินทาง ทานข้าว หรือกำลังเข้านอน ก็สามารถเปิดโทรศัพท์มือถือรวมทั้งเล่นสล็อตออนไลน์ได้แบบชิวๆสบายต่อการใช้แรงงานแบบสุดๆเลยล่ะขอรับ



 ฉันเองก็เป็นเพียงคนหนึ่งที่มองดูเกมพนันออนไลน์แบบผ่านๆไม่เคยคิดจะลองเล่นตั้งใจจริง กระทั่งได้มีโอกาสทดลองกับเว็บไซต์หนึ่งที่เพื่อนซี้แนะนำว่า รอยัล558 “เว็บนี้ดีสุดแล้ว ระบบดี มั่นคง ไม่คดโกง เล่นได้จริงจ่ายจริง” พอใช้ลองด้วยตัวเอง ก็จำต้องเห็นด้วยว่ามันแปลงมุมมองทั้งผองเกี่ยวกับโลกของเว็บพนันออนไลน์ไปแบบสิ้นเชิง
ฉันเองก็เป็นเพียงคนหนึ่งที่มองดูเกมพนันออนไลน์แบบผ่านๆไม่เคยคิดจะลองเล่นตั้งใจจริง กระทั่งได้มีโอกาสทดลองกับเว็บไซต์หนึ่งที่เพื่อนซี้แนะนำว่า รอยัล558 “เว็บนี้ดีสุดแล้ว ระบบดี มั่นคง ไม่คดโกง เล่นได้จริงจ่ายจริง” พอใช้ลองด้วยตัวเอง ก็จำต้องเห็นด้วยว่ามันแปลงมุมมองทั้งผองเกี่ยวกับโลกของเว็บพนันออนไลน์ไปแบบสิ้นเชิง ตอนต้นฉันเริ่มจากการเล่นบาคาร่าแบบทดลอง เนื่องจากยังไม่กล้าฝากเงินเยอะ แต่สิ่งที่ทำให้ติดใจตั้งแต่ทีแรกเป็น
ตอนต้นฉันเริ่มจากการเล่นบาคาร่าแบบทดลอง เนื่องจากยังไม่กล้าฝากเงินเยอะ แต่สิ่งที่ทำให้ติดใจตั้งแต่ทีแรกเป็น
 เว็บ pananthai มีทีมงานมืออาชีพที่รอให้ความช่วยเหลือเกื้อกูล ตอบคำถามเกี่ยวกับการ pananthai สมัคร การฝากถอน หรือโปรโมชั่นต่างๆตลอด 24 ชั่วโมง เพื่อผู้เล่นมั่นใจได้ว่ามีคนดูแลเสมอ
เว็บ pananthai มีทีมงานมืออาชีพที่รอให้ความช่วยเหลือเกื้อกูล ตอบคำถามเกี่ยวกับการ pananthai สมัคร การฝากถอน หรือโปรโมชั่นต่างๆตลอด 24 ชั่วโมง เพื่อผู้เล่นมั่นใจได้ว่ามีคนดูแลเสมอ ชนิดของเครดิตฟรีที่ dr888 มอบให้
ชนิดของเครดิตฟรีที่ dr888 มอบให้ dr888 มีเกมอะไรให้เล่นบ้าง?
dr888 มีเกมอะไรให้เล่นบ้าง? spbetflik88 สมัครง่ายด้านในไม่กี่ขั้นตอน
spbetflik88 สมัครง่ายด้านในไม่กี่ขั้นตอน spbetflik88 คือเว็บไซต์คาสิโนออนไลน์ที่ครบเครื่องทั้งยังด้านเกม ความปลอดภัย รวมทั้งโปรโมชั่นที่คุ้มค่า ถ้าหากคุณกำลังมองหาเว็บไซต์ที่สมัครง่าย เข้าระบบเร็วทันใจ และมี spbetflik88 เครดิตฟรี ให้ใช้งาน ที่นี่คือคำตอบที่ดีเยี่ยมที่สุด อย่าคอยช้า รีบสมัครรวมทั้งสัมผัสประสบการณ์การพนันที่เหนือระดับได้แล้ววันนี้
spbetflik88 คือเว็บไซต์คาสิโนออนไลน์ที่ครบเครื่องทั้งยังด้านเกม ความปลอดภัย รวมทั้งโปรโมชั่นที่คุ้มค่า ถ้าหากคุณกำลังมองหาเว็บไซต์ที่สมัครง่าย เข้าระบบเร็วทันใจ และมี spbetflik88 เครดิตฟรี ให้ใช้งาน ที่นี่คือคำตอบที่ดีเยี่ยมที่สุด อย่าคอยช้า รีบสมัครรวมทั้งสัมผัสประสบการณ์การพนันที่เหนือระดับได้แล้ววันนี้ เพราะเหตุใด fcharoenkit จึงเป็นตัวเลือกที่ใช่
เพราะเหตุใด fcharoenkit จึงเป็นตัวเลือกที่ใช่ ผู้เล่นสามารถเริ่ม fcharoenkit สมัคร ได้ด้วยตนเองผ่านระบบอัตโนมัติ เพียงแต่กรอกข้อมูลฐานรากไม่กี่ขั้นตอนก็พร้อมเข้าเล่นในทันที
ผู้เล่นสามารถเริ่ม fcharoenkit สมัคร ได้ด้วยตนเองผ่านระบบอัตโนมัติ เพียงแต่กรอกข้อมูลฐานรากไม่กี่ขั้นตอนก็พร้อมเข้าเล่นในทันที fcharoenkit เป็นเว็บคาสิโนออนไลน์ที่ครบเครื่องทั้งความสะดวก ปลอดภัย รวมทั้งโปรโมชั่นสุดคุ้ม ตั้งแต่ขั้นตอน
fcharoenkit เป็นเว็บคาสิโนออนไลน์ที่ครบเครื่องทั้งความสะดวก ปลอดภัย รวมทั้งโปรโมชั่นสุดคุ้ม ตั้งแต่ขั้นตอน  askyouwin888 vip จากวันนั้นจนกระทั่งวันนี้ ผมเล่นสล็อตมาหลายปี ผ่านทั้งยังช่วงที่ขาดทุนจนกระทั่งต้องการเลิก และก็ช่วงที่แจ็กพอตแตกกระทั่งยิ้มไม่หุบ และก็นี่คือประสบการณ์ตรงของผมที่อยากแชร์ให้ทุกคนได้รับรู้ว่า “เล่นสล็อตอย่างไรให้เพิ่มโอกาสชนะรางวัลใหญ่” โดยยิ่งไปกว่านั้นกับยุคที่มีระบบ ทดสอบเล่นสล็อตฟรี และก็ ฝากถอนออโต้ ที่ทำให้ทุกๆอย่างสบายขึ้นแบบสุดๆ
askyouwin888 vip จากวันนั้นจนกระทั่งวันนี้ ผมเล่นสล็อตมาหลายปี ผ่านทั้งยังช่วงที่ขาดทุนจนกระทั่งต้องการเลิก และก็ช่วงที่แจ็กพอตแตกกระทั่งยิ้มไม่หุบ และก็นี่คือประสบการณ์ตรงของผมที่อยากแชร์ให้ทุกคนได้รับรู้ว่า “เล่นสล็อตอย่างไรให้เพิ่มโอกาสชนะรางวัลใหญ่” โดยยิ่งไปกว่านั้นกับยุคที่มีระบบ ทดสอบเล่นสล็อตฟรี และก็ ฝากถอนออโต้ ที่ทำให้ทุกๆอย่างสบายขึ้นแบบสุดๆ เริ่มจาก การทดสอบเล่น
เริ่มจาก การทดสอบเล่น 
 บันเทิงใจกับสล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์จาก สล็อต168 ได้วันแล้ววันเล่า เว็บไซต์ตรงมาตรฐานระดับนานาชาติที่คุณจำต้องทดลอง สมัครเลย!
บันเทิงใจกับสล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์จาก สล็อต168 ได้วันแล้ววันเล่า เว็บไซต์ตรงมาตรฐานระดับนานาชาติที่คุณจำต้องทดลอง สมัครเลย!

 เล่นสล็อต Joker123 ที่ไหนดี? แชร์ประสบการณ์ตรง พร้อมคำเสนอแนะแบบไม่กั๊ก
เล่นสล็อต Joker123 ที่ไหนดี? แชร์ประสบการณ์ตรง พร้อมคำเสนอแนะแบบไม่กั๊ก กำลังมองหาเว็บไซต์เล่น
กำลังมองหาเว็บไซต์เล่น 
 รีวิวเว็บไซต์ลอตเตอรี่อันดับ 1 lotto432 จ่ายจริง จ่ายหนัก ไม่มีฉ้อฉล อัตราสูงสุดในประเทศ
รีวิวเว็บไซต์ลอตเตอรี่อันดับ 1 lotto432 จ่ายจริง จ่ายหนัก ไม่มีฉ้อฉล อัตราสูงสุดในประเทศ ย้อนกลับไปเมื่อปีก่อน ผมยังคงเป็นหนึ่งในผู้ที่ซื้อหวยแบบเดิมๆคือส่งโพยกับเจ้ามือแถวบ้าน มีอีกทั้งเลขกลั้น จ่ายครึ่ง บางเวลาก็จำเป็นต้องลุ้นว่าเงินจะได้ไหมหลังลอตเตอรี่ออก บางงวดก็โดนเบี้ยวเฉยๆไร้เหตุผลอะไรให้โกรธโดยทันที เพียงแค่ต้องยอมเพราะว่า เจ้าใหญ่ในพื้นที่
ย้อนกลับไปเมื่อปีก่อน ผมยังคงเป็นหนึ่งในผู้ที่ซื้อหวยแบบเดิมๆคือส่งโพยกับเจ้ามือแถวบ้าน มีอีกทั้งเลขกลั้น จ่ายครึ่ง บางเวลาก็จำเป็นต้องลุ้นว่าเงินจะได้ไหมหลังลอตเตอรี่ออก บางงวดก็โดนเบี้ยวเฉยๆไร้เหตุผลอะไรให้โกรธโดยทันที เพียงแค่ต้องยอมเพราะว่า เจ้าใหญ่ในพื้นที่

 เล่นสล็อต777 แบบเว้นรอบดีหรือไหม? หรือการสปินต่อเนื่องจะดีกว่า? มาหาคำตอบกัน!
เล่นสล็อต777 แบบเว้นรอบดีหรือไหม? หรือการสปินต่อเนื่องจะดีกว่า? มาหาคำตอบกัน! ผมชูให้เป็นไอเดียการเล่นเกมสล็อต777 ที่น่าสนใจที่สุดประจำปีนี้เลยก็ว่าได้ กับการเล่นสล็อต777แบบเว้นรอบและแบบสม่ำเสมอที่มือโปรนิยมใช้งานกันทั้งสองแบบ แล้วแบบไหนดียิ่งกว่ากันล่ะ? ในฐานะที่ผมเป็นนักตรวจตัวยงอยู่แล้ว ผมเคยใช้เทคนิคการเล่นทั้งยัง 2 แบบมาแล้วขอรับ ถ้าเกิดให้ผมเปรียบเทียบว่าแบบไหนดีมากกว่า มันก็จะเป็นการให้ร้ายนึกเห็นมากเกินความจำเป็น โดยเหตุนี้ ผมจะพาคุณมาสำรวจและมองหาไม่เหมือนกันไปพร้อมๆกันดีกว่า ชอบแบบไหนก็เลือกเองได้ตามใจแน่นอน ฝ่ากัน!
ผมชูให้เป็นไอเดียการเล่นเกมสล็อต777 ที่น่าสนใจที่สุดประจำปีนี้เลยก็ว่าได้ กับการเล่นสล็อต777แบบเว้นรอบและแบบสม่ำเสมอที่มือโปรนิยมใช้งานกันทั้งสองแบบ แล้วแบบไหนดียิ่งกว่ากันล่ะ? ในฐานะที่ผมเป็นนักตรวจตัวยงอยู่แล้ว ผมเคยใช้เทคนิคการเล่นทั้งยัง 2 แบบมาแล้วขอรับ ถ้าเกิดให้ผมเปรียบเทียบว่าแบบไหนดีมากกว่า มันก็จะเป็นการให้ร้ายนึกเห็นมากเกินความจำเป็น โดยเหตุนี้ ผมจะพาคุณมาสำรวจและมองหาไม่เหมือนกันไปพร้อมๆกันดีกว่า ชอบแบบไหนก็เลือกเองได้ตามใจแน่นอน ฝ่ากัน!
 sawan888 ที่ไม่ซ้ำใคร ทุกการหมุนเป็นจังหวะ
sawan888 ที่ไม่ซ้ำใคร ทุกการหมุนเป็นจังหวะ

 แถมยังมี ส่วนลด ให้เลือกอีก ยกตัวอย่างเช่น ถ้าเลือกแบบส่วนลด ก็จะได้ลดตอนซื้อ แต่ว่าหากเลือกแบบ จ่ายเต็มไม่ลด ก็จะได้อัตราจ่ายสูงสุด เรียกว่าเลือกได้ตามใจเลย
แถมยังมี ส่วนลด ให้เลือกอีก ยกตัวอย่างเช่น ถ้าเลือกแบบส่วนลด ก็จะได้ลดตอนซื้อ แต่ว่าหากเลือกแบบ จ่ายเต็มไม่ลด ก็จะได้อัตราจ่ายสูงสุด เรียกว่าเลือกได้ตามใจเลย 1.อัตราจ่ายสูงกว่าแบบชัดแจ้ง ถูกครั้งเดียวคุ้มกว่าหลายงวด
1.อัตราจ่ายสูงกว่าแบบชัดแจ้ง ถูกครั้งเดียวคุ้มกว่าหลายงวด ช่วงแรกที่ได้ยินคำว่า บาคาร่า ฉันยังไม่รู้เรื่องด้วยซ้ำว่ามันคือเกมอะไร รู้เพียงแค่ว่ามีคนพูดถึงกันมากมายในโลกออนไลน์ บ้างก็กล่าวว่า “เล่นง่าย เสมือนป๊อกกระเด้ง” บ้างก็บอกว่า “เป็นเกมของคนมีแนวทาง”
ช่วงแรกที่ได้ยินคำว่า บาคาร่า ฉันยังไม่รู้เรื่องด้วยซ้ำว่ามันคือเกมอะไร รู้เพียงแค่ว่ามีคนพูดถึงกันมากมายในโลกออนไลน์ บ้างก็กล่าวว่า “เล่นง่าย เสมือนป๊อกกระเด้ง” บ้างก็บอกว่า “เป็นเกมของคนมีแนวทาง” จนวันหนึ่ง เพื่อนฝูงเสนอแนะให้ทดลองสมัครเล่นผ่านเว็บไซต์ตรงที่เขาใช้อยู่ ด้วยเหตุว่าเป็นเว็บไซต์ศูนย์รวมค่ายบาคาร่าไว้ครบ — ทั้งยัง SA Gaming, Sexy Baccarat, Evolution,
จนวันหนึ่ง เพื่อนฝูงเสนอแนะให้ทดลองสมัครเล่นผ่านเว็บไซต์ตรงที่เขาใช้อยู่ ด้วยเหตุว่าเป็นเว็บไซต์ศูนย์รวมค่ายบาคาร่าไว้ครบ — ทั้งยัง SA Gaming, Sexy Baccarat, Evolution, 

 ถ้าจะเล่น
ถ้าจะเล่น พูดตรงๆเลยนะครับว่า ผมเคยผ่านช่วงลองผิดลองถูกมาก่อน โดนเว็บล็อกยูส โดนปรับ RTP เกม เล่นสล็อตเครดิตฟรียังไงก็ไม่แตก ถึงขั้นสงสัยว่าระบบมันล็อกไว้รึเปล่า จนถึงวันหนึ่งมีคนแนะนำให้ทดลองเว็บไซต์ชื่อ สล็อต168 ฟังชื่อก็แปลกหูดีครับผม ราวกับชื่อร้านอาหารจีน 555 แต่พอลองเข้าไปเล่นจริง… เปลี่ยนเป็นว่าผมไม่ได้อยากแปรไปเว็บอื่นอีกเลย
พูดตรงๆเลยนะครับว่า ผมเคยผ่านช่วงลองผิดลองถูกมาก่อน โดนเว็บล็อกยูส โดนปรับ RTP เกม เล่นสล็อตเครดิตฟรียังไงก็ไม่แตก ถึงขั้นสงสัยว่าระบบมันล็อกไว้รึเปล่า จนถึงวันหนึ่งมีคนแนะนำให้ทดลองเว็บไซต์ชื่อ สล็อต168 ฟังชื่อก็แปลกหูดีครับผม ราวกับชื่อร้านอาหารจีน 555 แต่พอลองเข้าไปเล่นจริง… เปลี่ยนเป็นว่าผมไม่ได้อยากแปรไปเว็บอื่นอีกเลย
 จุดเริ่มต้นของผมกับโลกของ
จุดเริ่มต้นของผมกับโลกของ  ผมเลยกลับไปใช้โหมด “ทดสอบเล่นฟรี” เพื่อเล่าเรียนจังหวะการหมุน, ระบบโบนัส, และความถี่ของการเข้าสู่รอบฟรีสปิน
ผมเลยกลับไปใช้โหมด “ทดสอบเล่นฟรี” เพื่อเล่าเรียนจังหวะการหมุน, ระบบโบนัส, และความถี่ของการเข้าสู่รอบฟรีสปิน
 สล็อตเครดิตฟรี ฝากถอน AUTO ไม่มีอย่างต่ำ รองรับทุกแพลตฟอร์ม เล่นได้ทุกๆที่ ตลอดเวลา
สล็อตเครดิตฟรี ฝากถอน AUTO ไม่มีอย่างต่ำ รองรับทุกแพลตฟอร์ม เล่นได้ทุกๆที่ ตลอดเวลา จนกระทั่งวันหนึ่ง ผมได้ลองเล่นกับเว็บ สล็อต168 แล้วก็จำต้องบอกเลยว่า “แปลงมุมมองที่มีต่อเกมสล็อตออนไลน์ไปทั้งปวง” เพราะทุกสิ่งทุกอย่างตั้งแต่ระบบการเล่นไปจนถึงการฝากถอนมัน อัตโนมัติและก็ง่ายอย่างยิ่งไม่ต้องรอแอดไม่น ไม่ต้องส่งสลิป แล้วก็ที่สำคัญคือ ไม่มีอย่างน้อยเลยแม้กระทั้งบาทเดียว
จนกระทั่งวันหนึ่ง ผมได้ลองเล่นกับเว็บ สล็อต168 แล้วก็จำต้องบอกเลยว่า “แปลงมุมมองที่มีต่อเกมสล็อตออนไลน์ไปทั้งปวง” เพราะทุกสิ่งทุกอย่างตั้งแต่ระบบการเล่นไปจนถึงการฝากถอนมัน อัตโนมัติและก็ง่ายอย่างยิ่งไม่ต้องรอแอดไม่น ไม่ต้องส่งสลิป แล้วก็ที่สำคัญคือ ไม่มีอย่างน้อยเลยแม้กระทั้งบาทเดียว ก่อนหน้านี้ที่ผ่านมาผมเป็นคนที่ชอบเล่น happy168 อยู่แล้ว แต่จะเล่นทีไรก็ต้องคอยเช็กเวลา เพราะบางเว็บไซต์ระบบช้า หรือฝากเงินต้องรอแอดมินอนุมัตำหนิ บางครั้งอยากถอนเพียงแค่ 50 บาท ก็ทำไม่ได้น่าฟังติด “ขั้นต่ำ 100” บ้าง หรือบางโอกาสระบบถอนก็ล่มในช่วงคนเล่นมาก
ก่อนหน้านี้ที่ผ่านมาผมเป็นคนที่ชอบเล่น happy168 อยู่แล้ว แต่จะเล่นทีไรก็ต้องคอยเช็กเวลา เพราะบางเว็บไซต์ระบบช้า หรือฝากเงินต้องรอแอดมินอนุมัตำหนิ บางครั้งอยากถอนเพียงแค่ 50 บาท ก็ทำไม่ได้น่าฟังติด “ขั้นต่ำ 100” บ้าง หรือบางโอกาสระบบถอนก็ล่มในช่วงคนเล่นมาก
 PG การันตีแตกหนักทุกเกม ทดสอบเล่นสล็อตได้ทุกค่าย ลงทะเบียนเป็นสมาชิกฟรี!
PG การันตีแตกหนักทุกเกม ทดสอบเล่นสล็อตได้ทุกค่าย ลงทะเบียนเป็นสมาชิกฟรี! ผมยังจำวันแรกที่ตกลงใจลอง “เล่นสล็อตออนไลน์” ได้ดี มันเริ่มจากความสงสัยเล็กๆว่าเกมเหล่านี้เล่นอย่างไร เพราะอะไรใครๆถึงกล่าวกันว่า PG แตกง่าย แตกหนัก เล่นแล้วได้เงินจริง ในช่วงเวลานั้นผมก็ยังไม่รู้จักหรอกว่า “แตกหนัก” นี่มันจะแค่ไหน จนได้ลองกับตนเอง รวมทั้งเปลี่ยนเป็นจุดเปลี่ยนที่ทำให้ผมเข้าใจว่า เกมสล็อตออนไลน์ โดยเฉพาะอย่างยิ่งค่าย PG SLOT มันมิได้มีดีเพียงแค่ภาพงาม แต่ว่ายังเต็มไปด้วยจังหวะสำหรับในการทำเงินจริงๆ
ผมยังจำวันแรกที่ตกลงใจลอง “เล่นสล็อตออนไลน์” ได้ดี มันเริ่มจากความสงสัยเล็กๆว่าเกมเหล่านี้เล่นอย่างไร เพราะอะไรใครๆถึงกล่าวกันว่า PG แตกง่าย แตกหนัก เล่นแล้วได้เงินจริง ในช่วงเวลานั้นผมก็ยังไม่รู้จักหรอกว่า “แตกหนัก” นี่มันจะแค่ไหน จนได้ลองกับตนเอง รวมทั้งเปลี่ยนเป็นจุดเปลี่ยนที่ทำให้ผมเข้าใจว่า เกมสล็อตออนไลน์ โดยเฉพาะอย่างยิ่งค่าย PG SLOT มันมิได้มีดีเพียงแค่ภาพงาม แต่ว่ายังเต็มไปด้วยจังหวะสำหรับในการทำเงินจริงๆ

 ด้วยเหตุผลดังกล่าว สล็อตออนไลน์ มันควบคุมได้อย่างไม่ต้องสงสัยครับผม คุณสามารถปฏิบัติตามกรรมวิธีการที่ผมจะชี้แนะต่อไปนี้ได้อย่างแน่แท้ จะมีวิธีการแบบไหนบ้าง มาดูกันเลยครับผม!
ด้วยเหตุผลดังกล่าว สล็อตออนไลน์ มันควบคุมได้อย่างไม่ต้องสงสัยครับผม คุณสามารถปฏิบัติตามกรรมวิธีการที่ผมจะชี้แนะต่อไปนี้ได้อย่างแน่แท้ จะมีวิธีการแบบไหนบ้าง มาดูกันเลยครับผม! 1. ประเมินผลแพ้ชนะให้เป็นตัวเลขด้านใน 20-40 สปิน
1. ประเมินผลแพ้ชนะให้เป็นตัวเลขด้านใน 20-40 สปิน • FS +1 ฟอร์มเริ่มมา: คุณสามารถเพิ่มเบท 20-25% 10-20 สปินได้ในทันที เน้นย้ำเล่นสั้น รอคอนเฟิร์มก่อน
• FS +1 ฟอร์มเริ่มมา: คุณสามารถเพิ่มเบท 20-25% 10-20 สปินได้ในทันที เน้นย้ำเล่นสั้น รอคอนเฟิร์มก่อน • FS -1 ฟอร์มลบ: ลดเบทลง 20-30% พัก 3-5 นาที หรือจะย้ายเกมก็ได้ เพื่อตัดการขาดทุนเร็ว
• FS -1 ฟอร์มลบ: ลดเบทลง 20-30% พัก 3-5 นาที หรือจะย้ายเกมก็ได้ เพื่อตัดการขาดทุนเร็ว พนันสนุกสนานฝากถอนสบายกับ โจ๊กเกอร์123 สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ลำดับที่หนึ่งในไทย เล่นเกมสาแก่ใจได้ทุกเมื่อเชื่อวัน!
พนันสนุกสนานฝากถอนสบายกับ โจ๊กเกอร์123 สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ลำดับที่หนึ่งในไทย เล่นเกมสาแก่ใจได้ทุกเมื่อเชื่อวัน! โจ๊กเกอร์123 ที่มาพร้อมโปรโมชั่นและกิจกรรมมากไม่น้อยเลยทีเดียวเยอะมาก รับเครดิตฟรีแบบฟินๆได้ตามอยาก สนุกสนานกับเกมที่ถูกใจได้ทุกวี่ทุกวันตลอด 1 วัน คุณจะสนุกสนานกับการพนันสล็อตเยอะขึ้นเรื่อยๆ มีแอดมินคอยดูแลตลอดเวลา และก็ที่สำคัญ ฝากถอนง่ายไม่มีอย่างน้อยอีกด้วย หากคุณไม่ได้อยากต้องการเสียโอกาสดีๆอย่างนี้ล่ะก็ สมัครสมาชิกใหม่กับเราได้แล้ววันนี้ สมัครฟรี ไร้ค่าขนบธรรมเนียมประเพณี คลิกเลย!
โจ๊กเกอร์123 ที่มาพร้อมโปรโมชั่นและกิจกรรมมากไม่น้อยเลยทีเดียวเยอะมาก รับเครดิตฟรีแบบฟินๆได้ตามอยาก สนุกสนานกับเกมที่ถูกใจได้ทุกวี่ทุกวันตลอด 1 วัน คุณจะสนุกสนานกับการพนันสล็อตเยอะขึ้นเรื่อยๆ มีแอดมินคอยดูแลตลอดเวลา และก็ที่สำคัญ ฝากถอนง่ายไม่มีอย่างน้อยอีกด้วย หากคุณไม่ได้อยากต้องการเสียโอกาสดีๆอย่างนี้ล่ะก็ สมัครสมาชิกใหม่กับเราได้แล้ววันนี้ สมัครฟรี ไร้ค่าขนบธรรมเนียมประเพณี คลิกเลย! Joker123auto เป็นยังไง? เพราะเหตุใดถึงได้รับความนิยมเยอะที่สุดเวลานี้
Joker123auto เป็นยังไง? เพราะเหตุใดถึงได้รับความนิยมเยอะที่สุดเวลานี้
 เหตุผลที่ควรที่จะเลือกเล่นกับ khumsup888
เหตุผลที่ควรที่จะเลือกเล่นกับ khumsup888 การ
การ  ระบบ royaleluxe ปลอดภัยขนาดไหน?
ระบบ royaleluxe ปลอดภัยขนาดไหน? ข้อสรุป — เริ่มกับ sing55 อย่างชาญฉลาด
ข้อสรุป — เริ่มกับ sing55 อย่างชาญฉลาด

 ข้อสรุปและชักชวนให้เริ่ม
ข้อสรุปและชักชวนให้เริ่ม


 สล็อตเครดิตฟรี ไม่ได้เก็บรวบรวมเพียงแค่ เกมมาก แม้กระนั้นเป็น เกมที่แตกง่ายจริง โดยเลือกเฉพาะเกมสล็อตเครดิตฟรี ที่มีค่า RTP สูง ฟรีสปินเข้าไว และโบนัสออกบ่อยมากจากค่ายสล็อตสุดยอด เป็นต้นว่า PG Soft, Pragmatic Play,
สล็อตเครดิตฟรี ไม่ได้เก็บรวบรวมเพียงแค่ เกมมาก แม้กระนั้นเป็น เกมที่แตกง่ายจริง โดยเลือกเฉพาะเกมสล็อตเครดิตฟรี ที่มีค่า RTP สูง ฟรีสปินเข้าไว และโบนัสออกบ่อยมากจากค่ายสล็อตสุดยอด เป็นต้นว่า PG Soft, Pragmatic Play,  ยิ่งกว่านั้นยังมีเกมใหม่อัปเดตทุกสัปดาห์ พร้อมระบบจัดอันดับเกม แตกหนักทุกวัน ที่ผู้เล่นใช้มองแนวโน้มก่อนวางเดิมพันได้อีกด้วย
ยิ่งกว่านั้นยังมีเกมใหม่อัปเดตทุกสัปดาห์ พร้อมระบบจัดอันดับเกม แตกหนักทุกวัน ที่ผู้เล่นใช้มองแนวโน้มก่อนวางเดิมพันได้อีกด้วย
 918kiss เล่นกับเว็บตรงแตกง่าย การลงทุนในความสนุกแล้วก็ช่องทางทำกำไร
918kiss เล่นกับเว็บตรงแตกง่าย การลงทุนในความสนุกแล้วก็ช่องทางทำกำไร การเลือกเล่นกับ เว็บไซต์ตรง allbet24hr แตกง่าย นับว่าเป็นสิ่งสำคัญ เนื่องจากว่าเว็บไซต์ตรงมีข้อดีหลายประการที่เว็บไซต์เอเย่นต์ทั่วไปไม่สามารถให้ได้ เริ่มจากความปลอดภัยรวมทั้งความมั่นคงยั่งยืนทางการเงิน คุณไม่ต้องวิตกกังวลหัวข้อการทุจริตหรือปัญหาที่เกิดขึ้นกับการถอนเงินช้า ด้วยเหตุว่าทุกกระบวนการทำธุรกรรมผ่านระบบออโต้ที่ล้ำยุคและมีประสิทธิภาพสูง เป็นลู่ทางที่หลักแหลมจริงๆสำหรับคนที่ต้องการความสนุกสนานร่าเริงแล้วก็โอกาสสร้างรายได้ไปพร้อมๆกัน ด้วยความปลอดภัยสูง ระบบการคลังมั่นคง โบนัสแล้วก็แจ็คพอตแตกง่าย รวมถึงเกมนานัปการสไตล์ที่ทำให้ทุกการเดิมพันมีความตื่นเต้นรวมทั้งท้า การลงทุนใน 918kiss จึงไม่ใช่แค่การพนัน แต่ว่าคือการลงทุนในความสนุกสนานและก็จังหวะด้านการเงินที่คุ้มค่า
การเลือกเล่นกับ เว็บไซต์ตรง allbet24hr แตกง่าย นับว่าเป็นสิ่งสำคัญ เนื่องจากว่าเว็บไซต์ตรงมีข้อดีหลายประการที่เว็บไซต์เอเย่นต์ทั่วไปไม่สามารถให้ได้ เริ่มจากความปลอดภัยรวมทั้งความมั่นคงยั่งยืนทางการเงิน คุณไม่ต้องวิตกกังวลหัวข้อการทุจริตหรือปัญหาที่เกิดขึ้นกับการถอนเงินช้า ด้วยเหตุว่าทุกกระบวนการทำธุรกรรมผ่านระบบออโต้ที่ล้ำยุคและมีประสิทธิภาพสูง เป็นลู่ทางที่หลักแหลมจริงๆสำหรับคนที่ต้องการความสนุกสนานร่าเริงแล้วก็โอกาสสร้างรายได้ไปพร้อมๆกัน ด้วยความปลอดภัยสูง ระบบการคลังมั่นคง โบนัสแล้วก็แจ็คพอตแตกง่าย รวมถึงเกมนานัปการสไตล์ที่ทำให้ทุกการเดิมพันมีความตื่นเต้นรวมทั้งท้า การลงทุนใน 918kiss จึงไม่ใช่แค่การพนัน แต่ว่าคือการลงทุนในความสนุกสนานและก็จังหวะด้านการเงินที่คุ้มค่า



















 ขอขอบคุณเว็บ
ขอขอบคุณเว็บ 



 การันตีเกมแตกหนักแตกจริง ทุนน้อยเล่นได้ มือใหม่เล่นดีแน่ๆ สล็อตเครดิตฟรี เป็นมากกว่าเว็บไซต์สล็อตออนไลน์ทั่วไป รับประกันทุกความประทับใจแน่ๆ ถ้าเกิดคุณพร้อมจะมาสนุกสนานกับเราแล้วล่ะก็ มาสมัครเป็นสมาชิกใหม่กับเราได้เลย ลงทะเบียนเป็นสมาชิกใหม่ฟรี ไร้ค่าประเพณีแน่นอน คลิกเลย!
การันตีเกมแตกหนักแตกจริง ทุนน้อยเล่นได้ มือใหม่เล่นดีแน่ๆ สล็อตเครดิตฟรี เป็นมากกว่าเว็บไซต์สล็อตออนไลน์ทั่วไป รับประกันทุกความประทับใจแน่ๆ ถ้าเกิดคุณพร้อมจะมาสนุกสนานกับเราแล้วล่ะก็ มาสมัครเป็นสมาชิกใหม่กับเราได้เลย ลงทะเบียนเป็นสมาชิกใหม่ฟรี ไร้ค่าประเพณีแน่นอน คลิกเลย! แค่เข้าเกมแล้วกดเล่นก็พอแล้ว ซึ่งมันไม่มีอะไรผิดหรือถูกเลยขอรับ โดยเหตุนั้น ในพาร์ทนี้ ผมจะมาเสนอแนะการเล่นเกม punpro777 แบบไม่ต้องพึ่งสูตรการเล่นให้กังวลเกินความจำเป็น มาทดลองเล่นแบบชิวๆแต่มีหลักการกันเลยนะครับ
แค่เข้าเกมแล้วกดเล่นก็พอแล้ว ซึ่งมันไม่มีอะไรผิดหรือถูกเลยขอรับ โดยเหตุนั้น ในพาร์ทนี้ ผมจะมาเสนอแนะการเล่นเกม punpro777 แบบไม่ต้องพึ่งสูตรการเล่นให้กังวลเกินความจำเป็น มาทดลองเล่นแบบชิวๆแต่มีหลักการกันเลยนะครับ
 • ตอบปัญหาผู้เล่นทุกสไตล์:
• ตอบปัญหาผู้เล่นทุกสไตล์:  หลายคนบางทีอาจสงสัยว่า เกม pg77 ของพวกเรานั้นเหมาะกับผู้เล่นใหม่หรือผู้เล่นมือโปรมากยิ่งกว่ากัน? ผมบอกเลยนะครับว่า นี่เป็นอีกคำถามที่ถามกันจำนวนไม่ใช่น้อยเพราะเหตุว่าหากคุณเข้ามาอ่านบทความเกี่ยวกับเกม pgslot อยู่เรื่อยๆก็มีทั้งการชี้แนะเกมที่เหมาะกับมือใหม่หรือมือโปรอยู่แล้ว เพราะฉะนั้น ถ้าเกิดให้ผมสรุปแบบง่ายๆก็คือ มันเหมาะกับนักพนันทุกคนทุกกลุ่มอยู่แล้ว ไม่ว่าคุณจะเป็นมือใหม่หรือมืออาชีพ คุณก็สามารถเข้ามาได้กำไรกับ pg77 ได้อย่างแน่นอนครับผม มาทดลองกันได้เลย!
หลายคนบางทีอาจสงสัยว่า เกม pg77 ของพวกเรานั้นเหมาะกับผู้เล่นใหม่หรือผู้เล่นมือโปรมากยิ่งกว่ากัน? ผมบอกเลยนะครับว่า นี่เป็นอีกคำถามที่ถามกันจำนวนไม่ใช่น้อยเพราะเหตุว่าหากคุณเข้ามาอ่านบทความเกี่ยวกับเกม pgslot อยู่เรื่อยๆก็มีทั้งการชี้แนะเกมที่เหมาะกับมือใหม่หรือมือโปรอยู่แล้ว เพราะฉะนั้น ถ้าเกิดให้ผมสรุปแบบง่ายๆก็คือ มันเหมาะกับนักพนันทุกคนทุกกลุ่มอยู่แล้ว ไม่ว่าคุณจะเป็นมือใหม่หรือมืออาชีพ คุณก็สามารถเข้ามาได้กำไรกับ pg77 ได้อย่างแน่นอนครับผม มาทดลองกันได้เลย!
 ยิ่งเมื่อเลือกเล่นกับ สล็อตxo เว็บไซต์ตรงฝากถอน ไม่มีขั้นต่ำ คุณจะได้สัมผัสประสบการณ์การพนันที่แตกต่างไปจากเดิม ด้วยเหตุว่าเว็บตรงพวกนี้คือ เว็บสล็อตลิขสิทธิ์แท้ ที่ผ่านการตรวจสอบ มีใบอนุญาตเปิดบริการอย่างแม่นยำ และ รับประกันการจ่ายจริงทุกยอด ไม่ว่าจะเล่นได้กำไรเท่าไรก็ถอนได้เต็ม
ยิ่งเมื่อเลือกเล่นกับ สล็อตxo เว็บไซต์ตรงฝากถอน ไม่มีขั้นต่ำ คุณจะได้สัมผัสประสบการณ์การพนันที่แตกต่างไปจากเดิม ด้วยเหตุว่าเว็บตรงพวกนี้คือ เว็บสล็อตลิขสิทธิ์แท้ ที่ผ่านการตรวจสอบ มีใบอนุญาตเปิดบริการอย่างแม่นยำ และ รับประกันการจ่ายจริงทุกยอด ไม่ว่าจะเล่นได้กำไรเท่าไรก็ถอนได้เต็ม • เครดิตฟรีสมาชิกใหม่
• เครดิตฟรีสมาชิกใหม่

 มือใหม่ต้องลอง ทุนน้อยต้องโดน pgslot เว็บตรงลำดับที่หนึ่งในเอเชีย เครดิตฟรีเพียบ ถอนง่ายไม่มีอย่างน้อย คลิกเลย!
มือใหม่ต้องลอง ทุนน้อยต้องโดน pgslot เว็บตรงลำดับที่หนึ่งในเอเชีย เครดิตฟรีเพียบ ถอนง่ายไม่มีอย่างน้อย คลิกเลย! เกมสล็อตยอดฮิตที่ปล่อยมานานแม้กระนั้นยังแตกหนักในปี 2025 มีอะไรบ้าง มาดูกัน!
เกมสล็อตยอดฮิตที่ปล่อยมานานแม้กระนั้นยังแตกหนักในปี 2025 มีอะไรบ้าง มาดูกัน!

 เล่น funny18 แล้วได้เงินจริงไหม?
เล่น funny18 แล้วได้เงินจริงไหม?
 • บริหารเงินทุนให้เด่นชัด: คุณสามารถแบ่งเงินทุนออกเป็นรอบได้เลยจ๊าครับผม ไม่จำเป็นต้องใช้เงินลงทุนที่มีในการเล่นสล็อตรอบเดียว ดังเช่นว่า บางครั้งอาจจะแบ่งเป็น 3 ส่วนสำหรับเล่น 3 รอบ และอย่าลืมกำหนดจุดขาดทุนแล้วก็กำไรที่อยากได้ด้วย
• บริหารเงินทุนให้เด่นชัด: คุณสามารถแบ่งเงินทุนออกเป็นรอบได้เลยจ๊าครับผม ไม่จำเป็นต้องใช้เงินลงทุนที่มีในการเล่นสล็อตรอบเดียว ดังเช่นว่า บางครั้งอาจจะแบ่งเป็น 3 ส่วนสำหรับเล่น 3 รอบ และอย่าลืมกำหนดจุดขาดทุนแล้วก็กำไรที่อยากได้ด้วย เว็บไซต์ตรงไม่ผ่านเอเย่นต์ขวัญใจชาวไทย พุซซี่888 เพียงแค่นั้น! มือใหม่เล่นได้ 100% โปรโมชั่นจัดเต็มแน่นอน สมัครเลย!
เว็บไซต์ตรงไม่ผ่านเอเย่นต์ขวัญใจชาวไทย พุซซี่888 เพียงแค่นั้น! มือใหม่เล่นได้ 100% โปรโมชั่นจัดเต็มแน่นอน สมัครเลย!

 หากแม้กระนั้นเป็นเรื่อง ความปลอดภัย ความสบายสบาย และก็ความยืดหยุ่น สำหรับการฝากเบิกเงิน และหนึ่งในเว็บไซต์ที่ตอบปัญหาเรื่องนี้ได้อย่างลงตัวก็คือ สล็อต888 เว็บตรงประสิทธิภาพที่มาพร้อมระบบ สล็อตวอเลท ไม่มีอย่างต่ำ ที่ให้โอกาสให้ผู้เล่นทุกคนสามารถเริ่มต้นลงทุนได้หากแม้มีเงินเพียงแค่ 1 บาท
หากแม้กระนั้นเป็นเรื่อง ความปลอดภัย ความสบายสบาย และก็ความยืดหยุ่น สำหรับการฝากเบิกเงิน และหนึ่งในเว็บไซต์ที่ตอบปัญหาเรื่องนี้ได้อย่างลงตัวก็คือ สล็อต888 เว็บตรงประสิทธิภาพที่มาพร้อมระบบ สล็อตวอเลท ไม่มีอย่างต่ำ ที่ให้โอกาสให้ผู้เล่นทุกคนสามารถเริ่มต้นลงทุนได้หากแม้มีเงินเพียงแค่ 1 บาท
 เลือกเว็บไซต์ 168slot อย่างไรให้มีคุณภาพและเล่นเกมได้แบบไม่จำเป็นที่จะต้องมาวิตกกังวล
เลือกเว็บไซต์ 168slot อย่างไรให้มีคุณภาพและเล่นเกมได้แบบไม่จำเป็นที่จะต้องมาวิตกกังวล และก็ได้รับสิทธิประโยชน์ที่ยอดเยี่ยมซึ่งต่างจากสล็อต agent เป็นเว็บไซต์ที่มาจากเว็บตรงอีกครั้ง ซึ่งจำนวนมากแล้วเป็นเว็บไซต์ขนาดเล็กระบบ server ไม่ค่อยเสถียรเว็บล่มบ่อยมาก ไม่มีแอดมินรอดูแล รวมทั้งความพร้อมด้านการเงินน้อยกว่า 168slot เว็บตรง
และก็ได้รับสิทธิประโยชน์ที่ยอดเยี่ยมซึ่งต่างจากสล็อต agent เป็นเว็บไซต์ที่มาจากเว็บตรงอีกครั้ง ซึ่งจำนวนมากแล้วเป็นเว็บไซต์ขนาดเล็กระบบ server ไม่ค่อยเสถียรเว็บล่มบ่อยมาก ไม่มีแอดมินรอดูแล รวมทั้งความพร้อมด้านการเงินน้อยกว่า 168slot เว็บตรง

 สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง ปลอดภัย 100% เว็บตรงที่คุณตามหาจำต้องเว็บไซต์นี้แค่นั้น มาลองกัน!
สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง ปลอดภัย 100% เว็บตรงที่คุณตามหาจำต้องเว็บไซต์นี้แค่นั้น มาลองกัน! จัดเต็มเกม ปั่นแตก66 มากกว่าที่ไหนๆกับสล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง เว็บตรงแท้ 100% อิมพอร์ตจาก ปั่นแตก66 มีเกมให้เลือกเล่นล้นหลามกว่า 1,000 เกม เล่นผ่านเว็บได้ทันที ไม่ต้องดาวน์โหลดหรือจัดตั้งให้ยุ่งยาก บันเทิงใจกับเกม ปั่นแตก66 ที่ใช่ในแบบที่คุณชอบได้ทันที โปรโมชั่นแล้วก็กิจกรรมจัดเต็ม รับเครดิตฟรีได้ตั้งแต่ลงทะเบียน สล็อตเครดิตฟรีล่าสุดในขณะนี้ แจกไม่ยั้งกันเลยล่ะขอรับ รับประกันทุกความชอบใจแน่นอน แม้คุณไม่ได้อยากต้องการพลาดโอกาสในการได้กำไรที่เหมาะสมที่สุดล่ะก็ ลงทะเบียนเป็นสมาชิก ปั่นแตก66 ใหม่กับพวกเรากันได้เลย ค้ำประกันทุกความสนุกสนานแล้วก็ความรื่นเริงใจแน่นอน เพราะนี่คือเว็บไซต์ตรงที่ตามหาแน่นอน!
จัดเต็มเกม ปั่นแตก66 มากกว่าที่ไหนๆกับสล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง เว็บตรงแท้ 100% อิมพอร์ตจาก ปั่นแตก66 มีเกมให้เลือกเล่นล้นหลามกว่า 1,000 เกม เล่นผ่านเว็บได้ทันที ไม่ต้องดาวน์โหลดหรือจัดตั้งให้ยุ่งยาก บันเทิงใจกับเกม ปั่นแตก66 ที่ใช่ในแบบที่คุณชอบได้ทันที โปรโมชั่นแล้วก็กิจกรรมจัดเต็ม รับเครดิตฟรีได้ตั้งแต่ลงทะเบียน สล็อตเครดิตฟรีล่าสุดในขณะนี้ แจกไม่ยั้งกันเลยล่ะขอรับ รับประกันทุกความชอบใจแน่นอน แม้คุณไม่ได้อยากต้องการพลาดโอกาสในการได้กำไรที่เหมาะสมที่สุดล่ะก็ ลงทะเบียนเป็นสมาชิก ปั่นแตก66 ใหม่กับพวกเรากันได้เลย ค้ำประกันทุกความสนุกสนานแล้วก็ความรื่นเริงใจแน่นอน เพราะนี่คือเว็บไซต์ตรงที่ตามหาแน่นอน! ฝึกฝนเพื่อให้มีสมาธิกล้วยๆก่อนเล่นเกมกับ pgslot ช่วยอะไรได้บ้าง?
ฝึกฝนเพื่อให้มีสมาธิกล้วยๆก่อนเล่นเกมกับ pgslot ช่วยอะไรได้บ้าง? ผมไม่ใช่ผู้ที่เพิ่งฝึกหัดเล่น
ผมไม่ใช่ผู้ที่เพิ่งฝึกหัดเล่น • ถ้าหลังจากเล่นแล้วมีกำไรเพิ่มเป็น 200–300 บาท ผมจะกันทุน 100 ไว้ และค้ากำไรไปซื้อฟรีสปิน เพื่อหวังคูณรางวัลก้อนโต
• ถ้าหลังจากเล่นแล้วมีกำไรเพิ่มเป็น 200–300 บาท ผมจะกันทุน 100 ไว้ และค้ากำไรไปซื้อฟรีสปิน เพื่อหวังคูณรางวัลก้อนโต
 สล็อต168 มอบโอกาสให้ผู้เล่นได้สัมผัสประสบการณ์การเล่นสล็อต มิติใหม่ เล่นได้ทุกเกมส์
สล็อต168 มอบโอกาสให้ผู้เล่นได้สัมผัสประสบการณ์การเล่นสล็อต มิติใหม่ เล่นได้ทุกเกมส์ tangtem168 ไม่ได้เป็นเพียงแค่เกมสล็อตปกติ แต่เป็น แพลตฟอร์มความสนุกสนานเต็มต้นแบบ ที่ผสมความเพลิดเพลิน ความท้า รวมทั้งโอกาสสำหรับการสร้างรายได้ไว้ภายในที่เดียว ผู้เล่นสามารถเลือกเกมได้หลายร้อยเกม พร้อมธีมที่นานาประการ ตั้งแต่แฟนตาซี การเสี่ยงอันตราย วัฒนธรรมทวีปเอเชีย ไปจนถึงสล็อตแนวล้ำยุค
tangtem168 ไม่ได้เป็นเพียงแค่เกมสล็อตปกติ แต่เป็น แพลตฟอร์มความสนุกสนานเต็มต้นแบบ ที่ผสมความเพลิดเพลิน ความท้า รวมทั้งโอกาสสำหรับการสร้างรายได้ไว้ภายในที่เดียว ผู้เล่นสามารถเลือกเกมได้หลายร้อยเกม พร้อมธีมที่นานาประการ ตั้งแต่แฟนตาซี การเสี่ยงอันตราย วัฒนธรรมทวีปเอเชีย ไปจนถึงสล็อตแนวล้ำยุค • ความปลอดภัย – ใช้ระบบเข้ารหัสข้อมูล
• ความปลอดภัย – ใช้ระบบเข้ารหัสข้อมูล โปรโมชั่นสุดพิเศษสำหรับสมาชิกใหม่ tangtem168
โปรโมชั่นสุดพิเศษสำหรับสมาชิกใหม่ tangtem168 สนุกกับเว็บไซต์ตรงแท้หนึ่งเดียวในไทย pgslot77 เว็บตรงไม่ผ่านเอเย่นต์ ไม่เป็นอันตราย ไว้วางใจได้แน่ๆ สมัครเลย!
สนุกกับเว็บไซต์ตรงแท้หนึ่งเดียวในไทย pgslot77 เว็บตรงไม่ผ่านเอเย่นต์ ไม่เป็นอันตราย ไว้วางใจได้แน่ๆ สมัครเลย! ความสนุกสนานของการเดิมพันกับ pgslot77 เป็นอย่างไรรู้ไหมครับ? มันคือความแน่ใจในความปลอดภัยและสมรรถนะของเว็บไซต์นี่แหละครับ พวกเรา ทางเข้าpg รหัสฟรี เป็นเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง มั่นอกมั่นใจในความปลอดภัยได้อย่างไม่ต้องสงสัย คุณสามารถมาบันเทิงใจกับเกมทางเข้าpgลิขสิทธิ์แท้เยอะแยะจากค่ายเกมชั้นแนวหน้าระดับโลกได้ทุกวี่วันตลอด 24 ชั่วโมง มีโปรโมชั่นและก็กิจกรรมเยอะแยะมากมายก่ายกอง รับเครดิตฟรีได้ไม่จำกัด ฝากถอนสบายและเร็วทันใจกับระบบฝากถอนออโต้ เชื่อมั่นกับทุกๆการพนันได้อย่างแน่แท้ครับ ถ้าหากคุณพร้อมจะมาบันเทิงใจกับพวกเราแล้วล่ะก็ สมัครเป็นสมาชิกใหม่ได้แล้ววันนี้ คลิกเลย!
ความสนุกสนานของการเดิมพันกับ pgslot77 เป็นอย่างไรรู้ไหมครับ? มันคือความแน่ใจในความปลอดภัยและสมรรถนะของเว็บไซต์นี่แหละครับ พวกเรา ทางเข้าpg รหัสฟรี เป็นเว็บไซต์ตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง มั่นอกมั่นใจในความปลอดภัยได้อย่างไม่ต้องสงสัย คุณสามารถมาบันเทิงใจกับเกมทางเข้าpgลิขสิทธิ์แท้เยอะแยะจากค่ายเกมชั้นแนวหน้าระดับโลกได้ทุกวี่วันตลอด 24 ชั่วโมง มีโปรโมชั่นและก็กิจกรรมเยอะแยะมากมายก่ายกอง รับเครดิตฟรีได้ไม่จำกัด ฝากถอนสบายและเร็วทันใจกับระบบฝากถอนออโต้ เชื่อมั่นกับทุกๆการพนันได้อย่างแน่แท้ครับ ถ้าหากคุณพร้อมจะมาบันเทิงใจกับพวกเราแล้วล่ะก็ สมัครเป็นสมาชิกใหม่ได้แล้ววันนี้ คลิกเลย!
 แต่ว่ายังมาพร้อมฟีพบร์ที่หลากหลาย ไม่ว่าจะเป็น เดิมพันไม่มีขั้นต่ำ, ระบบฝากถอนออโต้, เล่นได้ตลอด 24 ชั่วโมง,
แต่ว่ายังมาพร้อมฟีพบร์ที่หลากหลาย ไม่ว่าจะเป็น เดิมพันไม่มีขั้นต่ำ, ระบบฝากถอนออโต้, เล่นได้ตลอด 24 ชั่วโมง,  การเล่น บาคาร่า168 แบบไม่มีอย่างน้อย เดิมพันไม่มีอย่างน้อย เหมาะกับทุกงบประมาณ
การเล่น บาคาร่า168 แบบไม่มีอย่างน้อย เดิมพันไม่มีอย่างน้อย เหมาะกับทุกงบประมาณ • เข้าหน้าเว็บไซต์ sexyauto168
• เข้าหน้าเว็บไซต์ sexyauto168 • อย่ามองแต่ว่าเค้าไพ่: มือใหม่มักจะติดตามเค้าไพ่เยอะเกินไป ชี้แนะให้ท่านใช้เป็นแถวทางในการเล่นเท่านั้น แม้กระนั้นอย่าใช้เป็นตัวพิพากษาสำหรับการเลือกเดิมพันเด็ดขาด เชื่อเซ้นท์ของตัวเอง เลือกเดิมพันในรอบที่คุณเชื่อมั่นเท่านั้นก็พอครับ
• อย่ามองแต่ว่าเค้าไพ่: มือใหม่มักจะติดตามเค้าไพ่เยอะเกินไป ชี้แนะให้ท่านใช้เป็นแถวทางในการเล่นเท่านั้น แม้กระนั้นอย่าใช้เป็นตัวพิพากษาสำหรับการเลือกเดิมพันเด็ดขาด เชื่อเซ้นท์ของตัวเอง เลือกเดิมพันในรอบที่คุณเชื่อมั่นเท่านั้นก็พอครับ ลงทะเบียนสมัครสมาชิกใหม่กับเรา บาคาร่า พนันได้อย่างปลอดภัย เชื่อมั่นในทุกการเดิมพัน ฝากถอนง่าย คลิกเลย!
ลงทะเบียนสมัครสมาชิกใหม่กับเรา บาคาร่า พนันได้อย่างปลอดภัย เชื่อมั่นในทุกการเดิมพัน ฝากถอนง่าย คลิกเลย! ขอขอบพระคุณby web
ขอขอบพระคุณby web  • รองรับระบบอัตโนมัติทั้งผอง ไม่ต้องแจ้งสลิป
• รองรับระบบอัตโนมัติทั้งผอง ไม่ต้องแจ้งสลิป • กิจกรรมหมุนวงล้อชิงของขวัญ
• กิจกรรมหมุนวงล้อชิงของขวัญ
 ● ลอตเตอรี่ฮานอยอื่นๆ: รวมทั้ง สลากกินแบ่งฮานอย lotto ที่เชื่อมโยงกับแพลตฟอร์มออนไลน์ แล้วก็ ลอตเตอรี่ฮานอย ลอตเตอรี่ซอง ซึ่งเป็นแนวทางจากสำนักต่างๆเป็นต้นว่า หวยฮานอย หลายสํานัก ที่รวบรวมเลขเด็ดจากหลายแหล่ง ข้อแนะนำเป็น อย่าเชื่อแนวทางเพียงแหล่งเดียว แม้กระนั้นให้เปรียบเทียบกับ หวยฮานอย ย้อนหลัง หรือ ลอตเตอรี่ฮานอยvipย้อนหลัง เพื่อความแม่นยำ
● ลอตเตอรี่ฮานอยอื่นๆ: รวมทั้ง สลากกินแบ่งฮานอย lotto ที่เชื่อมโยงกับแพลตฟอร์มออนไลน์ แล้วก็ ลอตเตอรี่ฮานอย ลอตเตอรี่ซอง ซึ่งเป็นแนวทางจากสำนักต่างๆเป็นต้นว่า หวยฮานอย หลายสํานัก ที่รวบรวมเลขเด็ดจากหลายแหล่ง ข้อแนะนำเป็น อย่าเชื่อแนวทางเพียงแหล่งเดียว แม้กระนั้นให้เปรียบเทียบกับ หวยฮานอย ย้อนหลัง หรือ ลอตเตอรี่ฮานอยvipย้อนหลัง เพื่อความแม่นยำ ● การตรวจผลรวมทั้งถ่ายทอดสด: สำหรับ สลากกินแบ่งฮานอย วันนี้ สด หรือ สลากกินแบ่งฮานอย สด, สามารถมองผ่าน YouTube หรือ สลากกินแบ่งฮานอย #ถ่ายทอดสดวันนี้ แม้ปรารถนา ลอตเตอรี่ฮานอย vip วันนี้สด หรือ สลากกินแบ่งฮานอย vipวันนี้สด, ชี้แนะติดตามวิถีทางอย่าง หวยฮานอย facebook ที่อัปเดตเรียลไทม์ ตัวอย่างผลล่าสุดจากวันที่ 24 ก.ค. 2568: ลอตเตอรี่ฮานอยvip เลข 4 ตัว 5823, 3 ตัว 823
● การตรวจผลรวมทั้งถ่ายทอดสด: สำหรับ สลากกินแบ่งฮานอย วันนี้ สด หรือ สลากกินแบ่งฮานอย สด, สามารถมองผ่าน YouTube หรือ สลากกินแบ่งฮานอย #ถ่ายทอดสดวันนี้ แม้ปรารถนา ลอตเตอรี่ฮานอย vip วันนี้สด หรือ สลากกินแบ่งฮานอย vipวันนี้สด, ชี้แนะติดตามวิถีทางอย่าง หวยฮานอย facebook ที่อัปเดตเรียลไทม์ ตัวอย่างผลล่าสุดจากวันที่ 24 ก.ค. 2568: ลอตเตอรี่ฮานอยvip เลข 4 ตัว 5823, 3 ตัว 823
 เล่น สล็อต แบบสายทุนน้อย ควรที่จะเลือกเล่นอย่างไร?
เล่น สล็อต แบบสายทุนน้อย ควรที่จะเลือกเล่นอย่างไร?
 จัดเต็มเกมสล็อตที่ยอดเยี่ยมจาก pgslot สล็อตเว็บไซต์ตรงแท้ 100% แตกหนักแตกจริง ถอนง่าย ทำเงินชิวๆฝ่าเลย!
จัดเต็มเกมสล็อตที่ยอดเยี่ยมจาก pgslot สล็อตเว็บไซต์ตรงแท้ 100% แตกหนักแตกจริง ถอนง่าย ทำเงินชิวๆฝ่าเลย! พร้อมจะมาสนุกกับพวกเรา สล็อตpg กันรึยังขอรับ? สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ที่มาแรงที่สุดขณะนี้ สล็อตเว็บตรงแท้ 100% จาก pgslot ที่มาพร้อมเกมสล็อตล้นหลาม เล่นผ่านเว็บไซต์ได้ทันที ไม่ต้องดาวน์โหลดหรือติดตั้งแอปพลิเคชัน สนุกกับเกมที่ใช่ในแบบที่ชอบได้ทุกวี่ทุกวัน รับรองเกมfullslotpgแตกง่าย แตกหนัก แตกจริง เบทต่ำสุด 1 บาทเพียงแค่นั้น งบน้อยเล่นได้ มือใหม่เล่นชิวแน่ๆ โปรโมชั่นและกิจกรรมจัดเต็มสุดๆฝากถอนง่าย ไม่มีอย่างน้อยในการฝากถอน สนุกสนานกับเกมที่ถูกใจได้ทุกเมื่อเชื่อวันตลอด 1 วัน มีแอดมินบริการด้วยนะครับ ถ้าหากคุณพึงพอใจล่ะก็ ลงทะเบียนเป็นสมาชิกใหม่แล้วรับเครดิตฟรีกันได้เลย อย่าคอยช้า ลุยเลย!
พร้อมจะมาสนุกกับพวกเรา สล็อตpg กันรึยังขอรับ? สล็อตเว็บไซต์ตรงไม่ผ่านเอเย่นต์ที่มาแรงที่สุดขณะนี้ สล็อตเว็บตรงแท้ 100% จาก pgslot ที่มาพร้อมเกมสล็อตล้นหลาม เล่นผ่านเว็บไซต์ได้ทันที ไม่ต้องดาวน์โหลดหรือติดตั้งแอปพลิเคชัน สนุกกับเกมที่ใช่ในแบบที่ชอบได้ทุกวี่ทุกวัน รับรองเกมfullslotpgแตกง่าย แตกหนัก แตกจริง เบทต่ำสุด 1 บาทเพียงแค่นั้น งบน้อยเล่นได้ มือใหม่เล่นชิวแน่ๆ โปรโมชั่นและกิจกรรมจัดเต็มสุดๆฝากถอนง่าย ไม่มีอย่างน้อยในการฝากถอน สนุกสนานกับเกมที่ถูกใจได้ทุกเมื่อเชื่อวันตลอด 1 วัน มีแอดมินบริการด้วยนะครับ ถ้าหากคุณพึงพอใจล่ะก็ ลงทะเบียนเป็นสมาชิกใหม่แล้วรับเครดิตฟรีกันได้เลย อย่าคอยช้า ลุยเลย!

 และก็มีความยั่งยืนและมั่นคงแปลงเป็นสิ่งจำเป็นมากยิ่งกว่าเดิม และหนึ่งในชื่อที่กำลังเดินทางมาแรงในปี 2025 ก็คือ hengjing168 เว็บตรงไม่ผ่านเอเย่นต์ เว็บไซต์ศูนย์รวมเกมสล็อตแตกบ่อย และก็อัปเดตทางเข้าใหม่ปัจจุบันให้เล่นได้แต่ละวันไม่มีสะดุด
และก็มีความยั่งยืนและมั่นคงแปลงเป็นสิ่งจำเป็นมากยิ่งกว่าเดิม และหนึ่งในชื่อที่กำลังเดินทางมาแรงในปี 2025 ก็คือ hengjing168 เว็บตรงไม่ผ่านเอเย่นต์ เว็บไซต์ศูนย์รวมเกมสล็อตแตกบ่อย และก็อัปเดตทางเข้าใหม่ปัจจุบันให้เล่นได้แต่ละวันไม่มีสะดุด

 สล็อต168 เว็บตรงไม่ผ่านเอเย่นต์เพียงอย่างเดียวในไทย เอนจอยกับการได้กำไรแบบจัดเต็ม ฝากถอนไม่มีขั้นต่ำ ลองเลย!
สล็อต168 เว็บตรงไม่ผ่านเอเย่นต์เพียงอย่างเดียวในไทย เอนจอยกับการได้กำไรแบบจัดเต็ม ฝากถอนไม่มีขั้นต่ำ ลองเลย! สล็อต ไม่ใช่เว็บไซต์ปกติ เพราะว่านี่คือแหล่งรวมเกมสล็อต888สายแตกหนักที่คนเล่นจริงบอกกันปากต่อปากว่า แตกทุกไม้ กำไรทุกวี่วัน” ฝากไม่มีอย่างต่ำ ถอนเยอะแค่ไหนก็ได้ ไม่มีกั๊ก พร้อมระบบออโต้สุดล้ำที่ทำงานเร็วไม่ถึง 5 วิ ไม่ต้องง้อแอดไม่น ไม่ต้องกลัวระบบพังทลาย ที่สำคัญคือมีเกมจากค่ายแท้ 100% ให้เลือกแบบทะลุพันเกมสล็อต888 ครบทุกแนว ทุกฟีล และโบนัสก็จัดหนักทุกยูส ไม่ใช่แค่สมาชิกใหม่
สล็อต ไม่ใช่เว็บไซต์ปกติ เพราะว่านี่คือแหล่งรวมเกมสล็อต888สายแตกหนักที่คนเล่นจริงบอกกันปากต่อปากว่า แตกทุกไม้ กำไรทุกวี่วัน” ฝากไม่มีอย่างต่ำ ถอนเยอะแค่ไหนก็ได้ ไม่มีกั๊ก พร้อมระบบออโต้สุดล้ำที่ทำงานเร็วไม่ถึง 5 วิ ไม่ต้องง้อแอดไม่น ไม่ต้องกลัวระบบพังทลาย ที่สำคัญคือมีเกมจากค่ายแท้ 100% ให้เลือกแบบทะลุพันเกมสล็อต888 ครบทุกแนว ทุกฟีล และโบนัสก็จัดหนักทุกยูส ไม่ใช่แค่สมาชิกใหม่ การฝากเงินสามารถทำได้ผ่านแอปแบงค์ชั้นนำของไทย หรือจะใช้วอลเลทก็รองรับหมด และแน่นอน… ไร้คุณค่าประเพณี ซ่อนเร้นแน่นอน ส่วนการถอนเงินก็ง่ายไม่แพ้กัน แค่กดถอนผ่านหน้าเว็บไซต์ เงินจะถูกโอนเข้าบัญชีของคุณโดยตรงข้างในไม่กี่วินาที
การฝากเงินสามารถทำได้ผ่านแอปแบงค์ชั้นนำของไทย หรือจะใช้วอลเลทก็รองรับหมด และแน่นอน… ไร้คุณค่าประเพณี ซ่อนเร้นแน่นอน ส่วนการถอนเงินก็ง่ายไม่แพ้กัน แค่กดถอนผ่านหน้าเว็บไซต์ เงินจะถูกโอนเข้าบัญชีของคุณโดยตรงข้างในไม่กี่วินาที ทุกเกมรองรับการเล่นสล็อต888ผ่านมือถือ แท็บเล็ต หรือคอมพิวเตอร์ โดยไม่ต้องดาวน์โหลดให้เปลืองความจำเครื่อง เพียงแค่คลิกก็เล่นได้โดยทันที ไม่หลุด ไม่ค้าง
ทุกเกมรองรับการเล่นสล็อต888ผ่านมือถือ แท็บเล็ต หรือคอมพิวเตอร์ โดยไม่ต้องดาวน์โหลดให้เปลืองความจำเครื่อง เพียงแค่คลิกก็เล่นได้โดยทันที ไม่หลุด ไม่ค้าง ถ้าเกิดอยากเล่นสล็อตแบบแตกง่าย ได้เงินจริง จำเป็นต้องไม่พลาด สล็อต888 เว็บไซต์ตรงตัวจริงที่กล้าแจก กล้าให้ แล้วก็กล้ารับผิดชอบ
ถ้าเกิดอยากเล่นสล็อตแบบแตกง่าย ได้เงินจริง จำเป็นต้องไม่พลาด สล็อต888 เว็บไซต์ตรงตัวจริงที่กล้าแจก กล้าให้ แล้วก็กล้ารับผิดชอบ เพราะเหตุไรต้องเลือกเล่นกับ
เพราะเหตุไรต้องเลือกเล่นกับ  และถ้าเกิดคุณยังไม่มีเทคนิค บาคาร่า168 ดีๆล่ะก็ มาลองใช้แนวทาง 3 ไม้ได้ด้วยเหมือนกัน ยังมีสูตรการเล่นดีๆอีกเยอะแยะรอคุณอยู่ ถ้าคุณอยากเอนจอยแล้วก็เป็นเศรษฐีแล้วในขณะนี้ มาสมัครเป็นสมาชิกใหม่ได้ทันที สมัครฟรี ไม่มีค่าประเพณี ลงทะเบียนเป็นสมาชิกเลย!
และถ้าเกิดคุณยังไม่มีเทคนิค บาคาร่า168 ดีๆล่ะก็ มาลองใช้แนวทาง 3 ไม้ได้ด้วยเหมือนกัน ยังมีสูตรการเล่นดีๆอีกเยอะแยะรอคุณอยู่ ถ้าคุณอยากเอนจอยแล้วก็เป็นเศรษฐีแล้วในขณะนี้ มาสมัครเป็นสมาชิกใหม่ได้ทันที สมัครฟรี ไม่มีค่าประเพณี ลงทะเบียนเป็นสมาชิกเลย!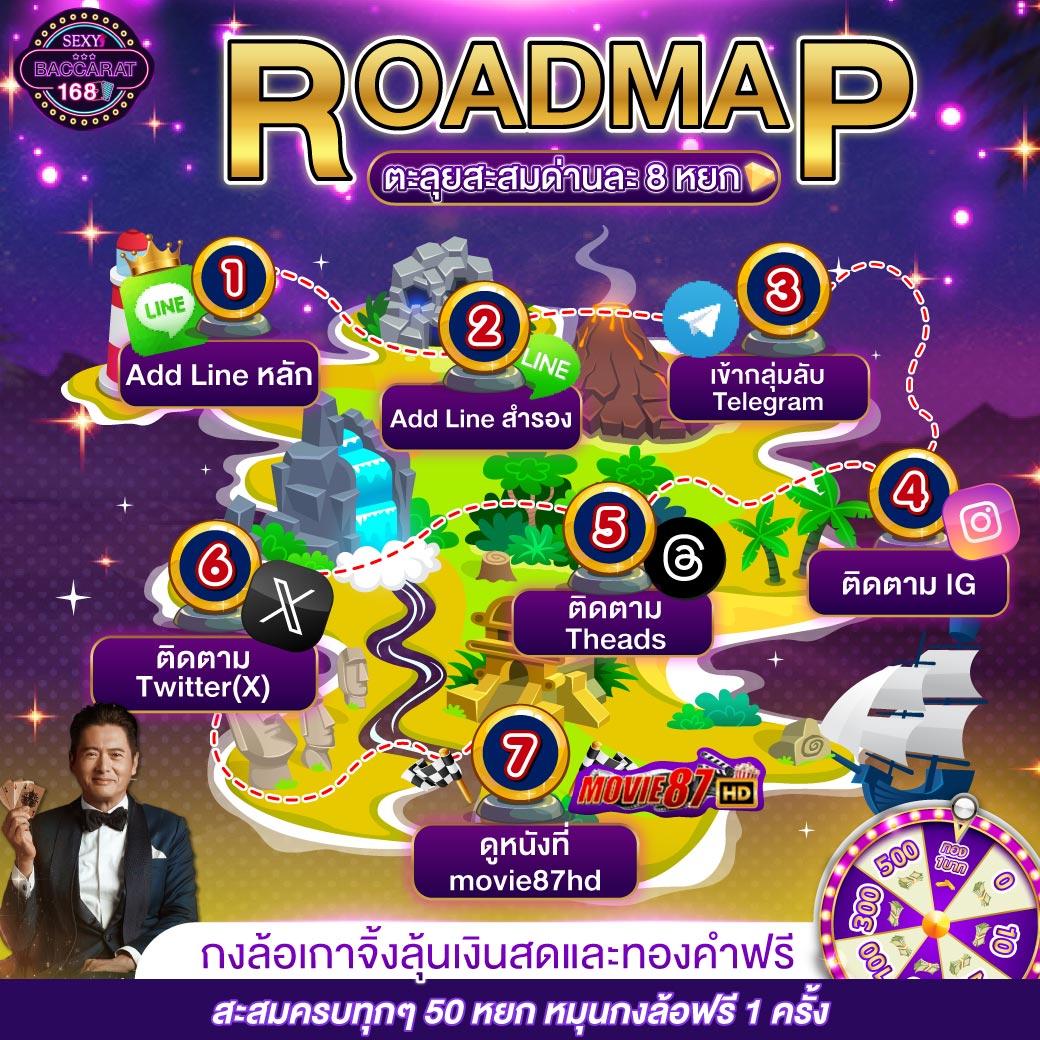

 บันเทิงใจกับเกมสล็อตที่ดีที่สุดบน joker123 ไม่พลาดทุกเกมดังที่ได้รับความนิยม แตกหนักแตกจริง 100% สมัครสมาชิกเลย!
บันเทิงใจกับเกมสล็อตที่ดีที่สุดบน joker123 ไม่พลาดทุกเกมดังที่ได้รับความนิยม แตกหนักแตกจริง 100% สมัครสมาชิกเลย! ช่วงเวลาสำหรับเพื่อการเล่น joker123auto ที่เหมาะสมที่สุดสำหรับมือโปร
ช่วงเวลาสำหรับเพื่อการเล่น joker123auto ที่เหมาะสมที่สุดสำหรับมือโปร

 ขวัญใจนักปั่นสล็อตรุ่นใหม่ สล็อต เว็บไซต์ตรงไม่ผ่านเอเย่นต์ API ระดับนานาชาติที่ยอดเยี่ยม สมัครสมาชิกใหม่ได้เลย!
ขวัญใจนักปั่นสล็อตรุ่นใหม่ สล็อต เว็บไซต์ตรงไม่ผ่านเอเย่นต์ API ระดับนานาชาติที่ยอดเยี่ยม สมัครสมาชิกใหม่ได้เลย! สล็อต เพราะเหตุใดสายปั่นสล็อตมือโปรถึงเลือกใช้บริการกับเรา!
สล็อต เพราะเหตุใดสายปั่นสล็อตมือโปรถึงเลือกใช้บริการกับเรา! • ทบทวนผลลัพธ์เสมอ: การทบทวนผลสรุปสำหรับในการเล่นเป็นรายสัปดาห์จะช่วยให้คุณแลเห็นภาพรวมของการเล่นสล็อตในแต่ละอาทิตย์เจริญขึ้น ถ้าหากมีการขาดทุนก็จะได้ปรับกลยุทธ์เล่นที่สมควรแล้วก็ดียิ่งขึ้นได้นั่นเองนะครับ
• ทบทวนผลลัพธ์เสมอ: การทบทวนผลสรุปสำหรับในการเล่นเป็นรายสัปดาห์จะช่วยให้คุณแลเห็นภาพรวมของการเล่นสล็อตในแต่ละอาทิตย์เจริญขึ้น ถ้าหากมีการขาดทุนก็จะได้ปรับกลยุทธ์เล่นที่สมควรแล้วก็ดียิ่งขึ้นได้นั่นเองนะครับ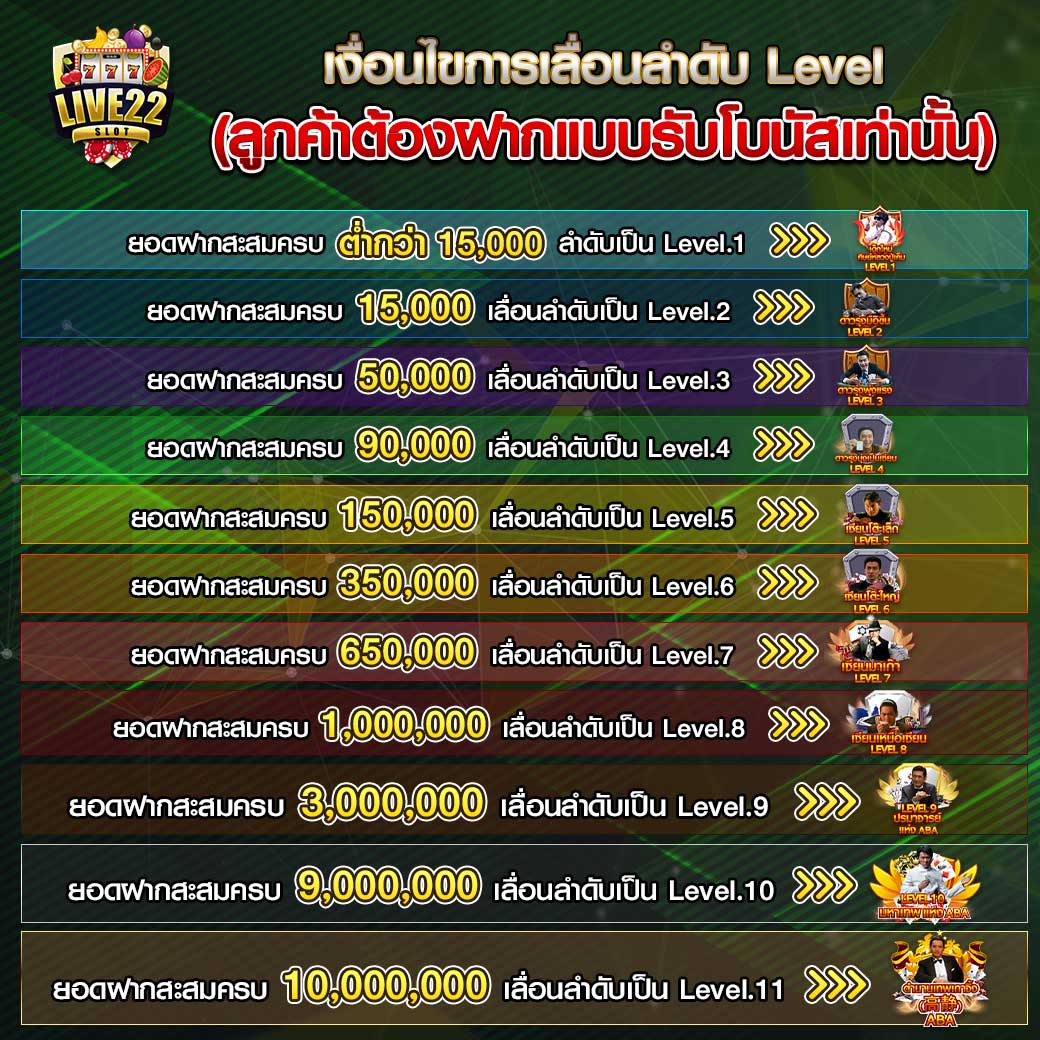
 เครดิตฟรี กลายเป็นชื่อที่ติดหูของเหล่าผู้เล่นเกมสล็อตออนไลน์ในยุคปัจจุบัน ด้วยความครบถ้วนบริบูรณ์ทั้งด้านเกมที่นานัปการ ระบบการเล่นgaojing888ที่ลื่นไหล แล้วก็โบนัสที่แตกง่ายแบบไม่มีกั๊ก ทำให้ที่นี่แปลงเป็น แหล่งรวมเกม สล็อตแตกหนัก ยอดนิยมสูงสุดจากนักปั่นสล็อตทั่วประเทศไทย โดยเฉพาะอย่างยิ่งค่ายเกมดังอย่าง พีจีสล็อต ที่ได้รับการรับรองว่าเป็นเกมแท้ 100% มีลิขสิทธิ์ถูกต้อง เล่นแล้วได้เงินจริง ไม่มีล็อกยูส
เครดิตฟรี กลายเป็นชื่อที่ติดหูของเหล่าผู้เล่นเกมสล็อตออนไลน์ในยุคปัจจุบัน ด้วยความครบถ้วนบริบูรณ์ทั้งด้านเกมที่นานัปการ ระบบการเล่นgaojing888ที่ลื่นไหล แล้วก็โบนัสที่แตกง่ายแบบไม่มีกั๊ก ทำให้ที่นี่แปลงเป็น แหล่งรวมเกม สล็อตแตกหนัก ยอดนิยมสูงสุดจากนักปั่นสล็อตทั่วประเทศไทย โดยเฉพาะอย่างยิ่งค่ายเกมดังอย่าง พีจีสล็อต ที่ได้รับการรับรองว่าเป็นเกมแท้ 100% มีลิขสิทธิ์ถูกต้อง เล่นแล้วได้เงินจริง ไม่มีล็อกยูส สล็อต888 เป็นเว็บตรงที่ให้บริการเกมสล็อตออนไลน์แบบครบวงจร สะสมเกมจากมากมายค่ายดังสุดยอด อาทิ PG SLOT, SLOTXO, JOKER GAMING, PRAGMATIC PLAY,
สล็อต888 เป็นเว็บตรงที่ให้บริการเกมสล็อตออนไลน์แบบครบวงจร สะสมเกมจากมากมายค่ายดังสุดยอด อาทิ PG SLOT, SLOTXO, JOKER GAMING, PRAGMATIC PLAY, 
 สล็อต168 เกมใหม่มาแรงอันดับ 1 แตกหนักจริง อัตราชนะมากที่สุด ฝาก-ถอน Auto รับประกัน เว็บไซต์ตรงเล่นง่าย
สล็อต168 เกมใหม่มาแรงอันดับ 1 แตกหนักจริง อัตราชนะมากที่สุด ฝาก-ถอน Auto รับประกัน เว็บไซต์ตรงเล่นง่าย


 แค่การเล่นเกมสนุกสิ่งเดียวแต่เป็นการเกื้อหนุนวิวัฒนาการของการเล่นเกมสล็อตออนไลน์ที่มีคุณภาพ admin มีความเป็นมืออาชีพ รวมทั้งยังทำให้การเล่นเกมได้เงินง่ายอีกด้วยมาบันเทิงใจไปร่วมกัน กับการเล่นเกมที่นี่มีมาตรฐานสูง และวันนี้พวกเราจะพาคุณไปดูว่า slot168 เหมาะสมสำหรับคนไหนกันแน่บ้าง
แค่การเล่นเกมสนุกสิ่งเดียวแต่เป็นการเกื้อหนุนวิวัฒนาการของการเล่นเกมสล็อตออนไลน์ที่มีคุณภาพ admin มีความเป็นมืออาชีพ รวมทั้งยังทำให้การเล่นเกมได้เงินง่ายอีกด้วยมาบันเทิงใจไปร่วมกัน กับการเล่นเกมที่นี่มีมาตรฐานสูง และวันนี้พวกเราจะพาคุณไปดูว่า slot168 เหมาะสมสำหรับคนไหนกันแน่บ้าง

 ในยุคที่เว็บไซต์สล็อตเกิดใหม่เยอะมาก การที่ joker123 ยังคงรักษาตำแหน่งเว็บอันดับที่หนึ่งได้นั้น ไม่ใช่ง่าย แต่ตรงนี้ทำเป็นเนื่องจากว่ารู้เรื่องรวมทั้งเอาใจใส่ผู้เล่นทุกคนอย่างแท้จริง นอกเหนือจากการที่จะอัปเดตเกมjoker123ใหม่ๆอย่างต่อเนื่องแล้ว ยังมีการพัฒนาระบบข้างหลังบ้านให้เสถียร ลื่นไหล ไม่มีสะดุด ทำให้ผู้เล่นมั่นใจว่าไม่ว่าจะเล่นช่วงไหน ก็ได้รับประสบการณ์เยี่ยมที่สุดเสมอ
ในยุคที่เว็บไซต์สล็อตเกิดใหม่เยอะมาก การที่ joker123 ยังคงรักษาตำแหน่งเว็บอันดับที่หนึ่งได้นั้น ไม่ใช่ง่าย แต่ตรงนี้ทำเป็นเนื่องจากว่ารู้เรื่องรวมทั้งเอาใจใส่ผู้เล่นทุกคนอย่างแท้จริง นอกเหนือจากการที่จะอัปเดตเกมjoker123ใหม่ๆอย่างต่อเนื่องแล้ว ยังมีการพัฒนาระบบข้างหลังบ้านให้เสถียร ลื่นไหล ไม่มีสะดุด ทำให้ผู้เล่นมั่นใจว่าไม่ว่าจะเล่นช่วงไหน ก็ได้รับประสบการณ์เยี่ยมที่สุดเสมอ ถ้าคุณกำลังมองหาเว็บไซต์joker123ที่ครบถ้วนบริบูรณ์ ตั้งแต่เกมหลากหลาย โปรโมชั่นจัดเต็ม ระบบเสถียร บริการดี และความปลอดภัยสูง joker123th คือคำตอบที่ไม่ควรมองข้าม
ถ้าคุณกำลังมองหาเว็บไซต์joker123ที่ครบถ้วนบริบูรณ์ ตั้งแต่เกมหลากหลาย โปรโมชั่นจัดเต็ม ระบบเสถียร บริการดี และความปลอดภัยสูง joker123th คือคำตอบที่ไม่ควรมองข้าม ขอขอบคุณอ้างอิง
ขอขอบคุณอ้างอิง  สล็อต 918kiss สล็อตเว็บไซต์ตรง เว็บใหญ่ที่สุดในไทย รวมเกมแตกง่าย ทดลองฟรี พร้อมโปรคุ้มทุกยูส
สล็อต 918kiss สล็อตเว็บไซต์ตรง เว็บใหญ่ที่สุดในไทย รวมเกมแตกง่าย ทดลองฟรี พร้อมโปรคุ้มทุกยูส ถ้าหากอยากเล่นสล็อต 918kiss แท้ ให้แตกง่ายได้เงินจริง allbet24hr เป็นคำตอบ
ถ้าหากอยากเล่นสล็อต 918kiss แท้ ให้แตกง่ายได้เงินจริง allbet24hr เป็นคำตอบ ยิ่งไปกว่านั้น โปรโมชั่นที่แจกทุกวี่วันโดยไม่มีเงื่อนไขยุ่งยาก ยังเป็นตัวช่วยสำคัญให้อีกทั้งผู้เล่นใหม่และก็ผู้เล่นเดิมสามารถเริ่มต้นหรือกลับมาเล่นได้อย่างมั่นใจ แบบไม่จำเป็นที่จะต้องมาวิตกกังวลหรือกลุ้มใจว่าจะขาดทุนโดยไม่จำเป็น ที่สำคัญที่สุดเป็นการบริการ—ตรงนี้คุณจะไม่ถูกละทิ้ง เพราะเหตุว่าคณะทำงาน allbet24hr พร้อมตอบทุกปัญหาและช่วยเหลือคุณทุกเวลา
ยิ่งไปกว่านั้น โปรโมชั่นที่แจกทุกวี่วันโดยไม่มีเงื่อนไขยุ่งยาก ยังเป็นตัวช่วยสำคัญให้อีกทั้งผู้เล่นใหม่และก็ผู้เล่นเดิมสามารถเริ่มต้นหรือกลับมาเล่นได้อย่างมั่นใจ แบบไม่จำเป็นที่จะต้องมาวิตกกังวลหรือกลุ้มใจว่าจะขาดทุนโดยไม่จำเป็น ที่สำคัญที่สุดเป็นการบริการ—ตรงนี้คุณจะไม่ถูกละทิ้ง เพราะเหตุว่าคณะทำงาน allbet24hr พร้อมตอบทุกปัญหาและช่วยเหลือคุณทุกเวลา
 เปิดประวัติความเป็นมาเกม pgslot ตั้งแต่เป็นตู้เกมจนถึงโทรศัพท์มือถือที่แปลงวงการไปตลอดกาล
เปิดประวัติความเป็นมาเกม pgslot ตั้งแต่เป็นตู้เกมจนถึงโทรศัพท์มือถือที่แปลงวงการไปตลอดกาล

 การเข้าเล่นก็ง่ายอย่างยิ่ง ไม่ว่าจะผ่านแอปพลิเคชันหรือโปรแกรมเบราเซอร์ ก็สามารถเข้าระบบรวมทั้งเริ่มบันเทิงใจกับเกมสล็อตได้โดยทันที ใช้งานได้ทั้งยังบนระบบ iOS และก็ Android รองรับทุกอุปกรณ์ไม่ว่าจะเป็นโทรศัพท์เคลื่อนที่ แท็บเล็ต หรือคอมพิวเตอร์
การเข้าเล่นก็ง่ายอย่างยิ่ง ไม่ว่าจะผ่านแอปพลิเคชันหรือโปรแกรมเบราเซอร์ ก็สามารถเข้าระบบรวมทั้งเริ่มบันเทิงใจกับเกมสล็อตได้โดยทันที ใช้งานได้ทั้งยังบนระบบ iOS และก็ Android รองรับทุกอุปกรณ์ไม่ว่าจะเป็นโทรศัพท์เคลื่อนที่ แท็บเล็ต หรือคอมพิวเตอร์ สล็อตxo ทุนน้อยก็เล่นได้ ช่องทางแตกไม่แพ้ใคร
สล็อตxo ทุนน้อยก็เล่นได้ ช่องทางแตกไม่แพ้ใคร • อินเทอร์เฟซใช้งานง่าย เหมาะสมกับผู้เล่นทุกวัย แม้ไม่มีประสบการณ์ก็เล่นได้สบาย
• อินเทอร์เฟซใช้งานง่าย เหมาะสมกับผู้เล่นทุกวัย แม้ไม่มีประสบการณ์ก็เล่นได้สบาย
 • รวมเกม
• รวมเกม โปรโมชันสุดฮอตจาก สล็อต ที่คนใดเห็นก็จะต้องว้าว เนื่องจากว่าให้จริง แจกจริง แบบไม่ต้องลุ้น
โปรโมชันสุดฮอตจาก สล็อต ที่คนใดเห็นก็จะต้องว้าว เนื่องจากว่าให้จริง แจกจริง แบบไม่ต้องลุ้น การมีระบบระเบียบทดสอบเล่นสล็อตฟรีแบบมือโปร เป็นอีกหนึ่งเหตุผลที่ทำให้ temmax69 แปลงเป็นเว็บไซต์สล็อตที่ผู้เล่นวางใจรวมทั้งเลือกเล่นกันอย่างต่อเนื่อง เพราะว่าตรงนี้คุณไม่ต้องเสี่ยงก่อน ไม่ต้องลองผิดลองถูกกับเงินจริง แต่ว่าได้ลองทุกเกมที่อยากเล่นtemmax69ก่อน พร้อมเตรียมความพร้อมให้พร้อมก่อนลงสนามจริง
การมีระบบระเบียบทดสอบเล่นสล็อตฟรีแบบมือโปร เป็นอีกหนึ่งเหตุผลที่ทำให้ temmax69 แปลงเป็นเว็บไซต์สล็อตที่ผู้เล่นวางใจรวมทั้งเลือกเล่นกันอย่างต่อเนื่อง เพราะว่าตรงนี้คุณไม่ต้องเสี่ยงก่อน ไม่ต้องลองผิดลองถูกกับเงินจริง แต่ว่าได้ลองทุกเกมที่อยากเล่นtemmax69ก่อน พร้อมเตรียมความพร้อมให้พร้อมก่อนลงสนามจริง
 ส่งต่อความสนุกไปกับการพนันบน
ส่งต่อความสนุกไปกับการพนันบน  ผูกรวมทุกฟีพบร์สุดพิเศษบน เกมสล็อต ที่บอกเลยว่า ทำเงินได้โคตรไว!
ผูกรวมทุกฟีพบร์สุดพิเศษบน เกมสล็อต ที่บอกเลยว่า ทำเงินได้โคตรไว!
 สล็อต168 ทำไมต้องเลือก สล็อตเครดิตฟรี 50 รับรองเบอร์?
สล็อต168 ทำไมต้องเลือก สล็อตเครดิตฟรี 50 รับรองเบอร์? มองหาเว็บ hengjing168 ที่น่าไว้วางใจ ไม่เป็นอันตราย รวมทั้งมาพร้อมกับโปรโมชันสุดคุ้มอย่าง สล็อตเครดิตฟรี 50 การันตีเบอร์ อย่าลืมเลือกเล่นกับ เว็บตรง ไม่ผ่านเอเย่นต์ ที่มีระบบฝาก-ถอนแบบไม่มีอย่างต่ำ คุณจะได้สัมผัสประสบการณ์การเล่นสล็อตที่ทั้งสนุกสนาน คุ้ม และไร้ความกังวลใจในทุกๆการเดิมพัน
มองหาเว็บ hengjing168 ที่น่าไว้วางใจ ไม่เป็นอันตราย รวมทั้งมาพร้อมกับโปรโมชันสุดคุ้มอย่าง สล็อตเครดิตฟรี 50 การันตีเบอร์ อย่าลืมเลือกเล่นกับ เว็บตรง ไม่ผ่านเอเย่นต์ ที่มีระบบฝาก-ถอนแบบไม่มีอย่างต่ำ คุณจะได้สัมผัสประสบการณ์การเล่นสล็อตที่ทั้งสนุกสนาน คุ้ม และไร้ความกังวลใจในทุกๆการเดิมพัน เกมใหม่ชี้แนะระดูนี้จาก สล็อต168
เกมใหม่ชี้แนะระดูนี้จาก สล็อต168



 ปั่นสล็อต กับ Coming88 เว็บไซต์ สล็อตแตกง่ายโบนัสแตกบ่อยมากสุดในปี 2025
ปั่นสล็อต กับ Coming88 เว็บไซต์ สล็อตแตกง่ายโบนัสแตกบ่อยมากสุดในปี 2025 ปั่นสล็อต Coming88 คือคำตอบสำหรับนักพนันที่กำลังมองหาแพลตฟอร์มสล็อตออนไลน์ที่ทั้งสนุกสนานแล้วก็ทำกำไรได้จริงในปี 2025 ด้วยการเป็น สล็อตเว็บตรง ที่ไม่ผ่านเอเย่นต์ Coming88 เสนอประสบการณ์การเล่นที่ปลอดภัย โปร่งใส และก็เต็มไปด้วยจังหวะในการลุ้นรางวัลใหญ่ ไม่ว่าคุณจะเป็นมือใหม่หรือเซียนสล็อต Coming88 เป็นแพลตฟอร์มที่ได้รับความนิยมสำหรับการปั่นสล็อต พร้อมกลเม็ดและก็ข้อมูลสำคัญเพื่อเพิ่มช่องทางทำกำไร
ปั่นสล็อต Coming88 คือคำตอบสำหรับนักพนันที่กำลังมองหาแพลตฟอร์มสล็อตออนไลน์ที่ทั้งสนุกสนานแล้วก็ทำกำไรได้จริงในปี 2025 ด้วยการเป็น สล็อตเว็บตรง ที่ไม่ผ่านเอเย่นต์ Coming88 เสนอประสบการณ์การเล่นที่ปลอดภัย โปร่งใส และก็เต็มไปด้วยจังหวะในการลุ้นรางวัลใหญ่ ไม่ว่าคุณจะเป็นมือใหม่หรือเซียนสล็อต Coming88 เป็นแพลตฟอร์มที่ได้รับความนิยมสำหรับการปั่นสล็อต พร้อมกลเม็ดและก็ข้อมูลสำคัญเพื่อเพิ่มช่องทางทำกำไร เพราะเหตุใดต้องเลือกปั่นสล็อต Coming88
เพราะเหตุใดต้องเลือกปั่นสล็อต Coming88
 เดิมพันกีฬา: พนันบอล บาสเก็ตบอล แล้วก็กีฬายอดฮิตอื่นๆด้วยอัตราต่อรองที่แข่งได้.
เดิมพันกีฬา: พนันบอล บาสเก็ตบอล แล้วก็กีฬายอดฮิตอื่นๆด้วยอัตราต่อรองที่แข่งได้. กำหนดงบประมาณ: ตั้งเป้าหมายการเล่นแล้วก็จำนวนเงินที่พร้อมใช้ เพื่อควบคุมการเดิมพันอย่างมีวินัย.
กำหนดงบประมาณ: ตั้งเป้าหมายการเล่นแล้วก็จำนวนเงินที่พร้อมใช้ เพื่อควบคุมการเดิมพันอย่างมีวินัย. ขอขอบคุณมากอ้างอิง
ขอขอบคุณมากอ้างอิง  P2vvip: เว็บสล็อตออนไลน์ที่ครบวงจรแล้วก็ดีเยี่ยมที่สุดในปี 2025
P2vvip: เว็บสล็อตออนไลน์ที่ครบวงจรแล้วก็ดีเยี่ยมที่สุดในปี 2025 ลักษณะเด่นของ P2VVIP
ลักษณะเด่นของ P2VVIP สมัครแทงบอลออนไลน์ p2vvip p2vvips.com 22 พฤษภา 2567 Epifania เกมแทงบอลสุดมันส์ p2vvipไม่ล็อคยูส Top 79
สมัครแทงบอลออนไลน์ p2vvip p2vvips.com 22 พฤษภา 2567 Epifania เกมแทงบอลสุดมันส์ p2vvipไม่ล็อคยูส Top 79

 บทความนี้ผมจะพาไปดูว่า sretthi99 มีคุณลักษณะเด่นอะไร ทำไมถึงแปลงเป็น สล็อตออนไลน์ชั้น 1 ที่ผู้เล่นไว้เนื้อเชื่อใจ พร้อมแนะนำ สูตรสล็อต 2025 ที่ผมใช้จริง และก็เห็นผลจริง รวมทั้งบอกต่อโปรโมชั่น เชื้อเชิญเพื่อนฝูงรับเครดิตฟรี ที่สามารถกลายเป็นเงินจริงได้แบบไม่ต้องลงทุนเองด้วยขอรับ
บทความนี้ผมจะพาไปดูว่า sretthi99 มีคุณลักษณะเด่นอะไร ทำไมถึงแปลงเป็น สล็อตออนไลน์ชั้น 1 ที่ผู้เล่นไว้เนื้อเชื่อใจ พร้อมแนะนำ สูตรสล็อต 2025 ที่ผมใช้จริง และก็เห็นผลจริง รวมทั้งบอกต่อโปรโมชั่น เชื้อเชิญเพื่อนฝูงรับเครดิตฟรี ที่สามารถกลายเป็นเงินจริงได้แบบไม่ต้องลงทุนเองด้วยขอรับ เพราะเหตุใดถึงเป็นเว็บสล็อตที่มีคนเล่นมากที่สุดเวลานี้?
เพราะเหตุใดถึงเป็นเว็บสล็อตที่มีคนเล่นมากที่สุดเวลานี้?
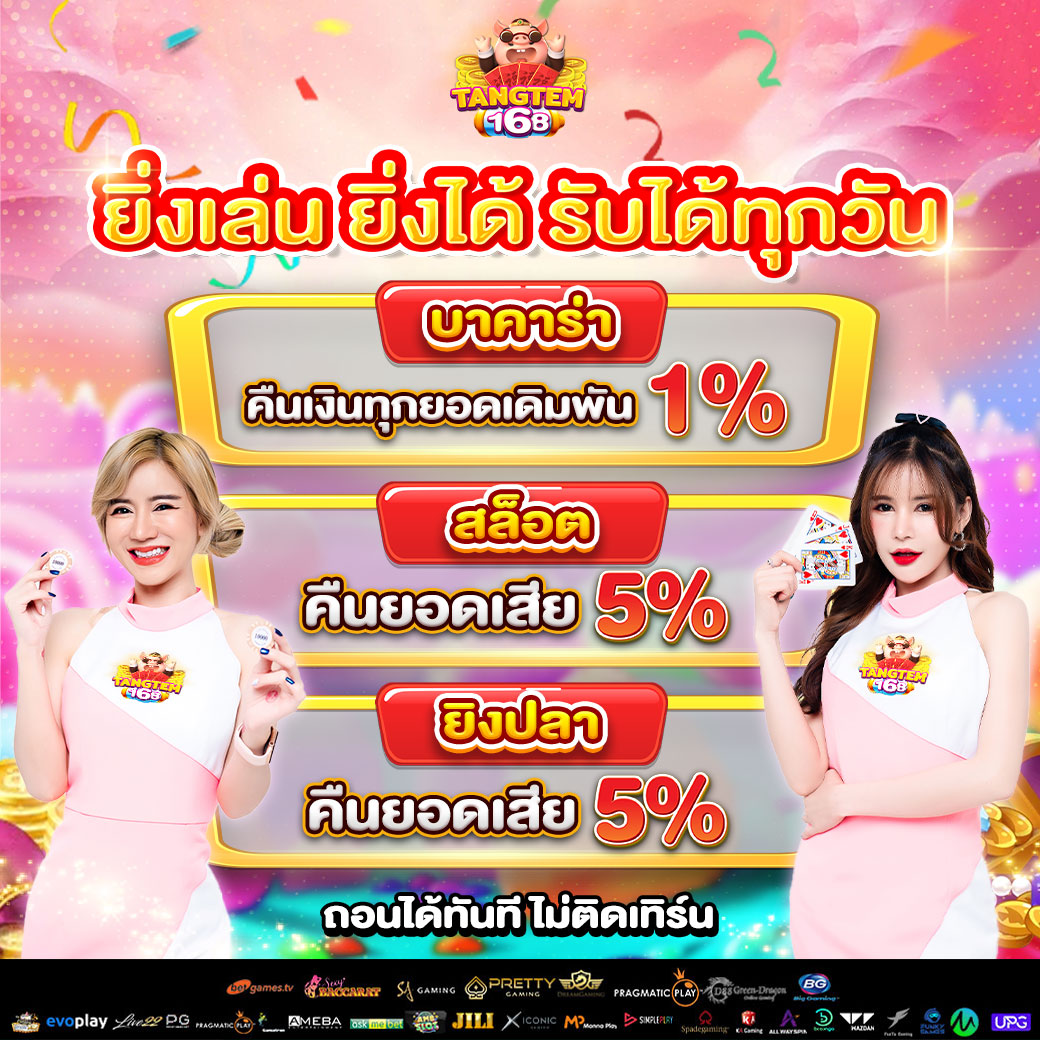
 ปากทางเข้าเดียว ครบทุกค่าย จบในยูสเดียว tangtem168
ปากทางเข้าเดียว ครบทุกค่าย จบในยูสเดียว tangtem168
 สล็อต คาสิโน เว็บไซต์ตรง ยอดฮิต ที่สุดของความมั่นคงยั่งยืน แตกจริง จ่ายชัวร์ ครบทุกเกมในเว็บเดียว
สล็อต คาสิโน เว็บไซต์ตรง ยอดฮิต ที่สุดของความมั่นคงยั่งยืน แตกจริง จ่ายชัวร์ ครบทุกเกมในเว็บเดียว • หน้าเว็บโหลดไว ใช้งานง่าย ไม่สลับซับซ้อน
• หน้าเว็บโหลดไว ใช้งานง่าย ไม่สลับซับซ้อน เว็บสล็อตตรง100% สล็อต 188betth.win 9 JUN 2568 Nigel casino online 188betth เว็บไหนดี Top 74
เว็บสล็อตตรง100% สล็อต 188betth.win 9 JUN 2568 Nigel casino online 188betth เว็บไหนดี Top 74
 ในยุคที่ทุกวินาทีมีค่า happy168 จึงตั้งมั่นสร้างแพลตฟอร์มเกมสล็อตออนไลน์ที่ไม่ได้แค่ให้คุณเล่น แม้กระนั้นพาคุณทำเงินอย่างชาญฉลาด เว็บไซต์นี้เน้นที่การให้บริการแบบสุจริตใจ เกมที่คัดสรรมาเฉพาะตัวแตกไว ฟีพบร์ล้ำ และโบนัสแจกจริงวันแล้ววันเล่า ไม่ต้องหวังพึ่งดวงอย่างเดียวอีกต่อไป เนื่องจากระบบหลังบ้านของ สล็อต168 ถูกวางแบบมาเพื่อ “ช่วยเหลือ” ให้ผู้เล่นมีโอกาสสร้างรายได้เยอะที่สุด
ในยุคที่ทุกวินาทีมีค่า happy168 จึงตั้งมั่นสร้างแพลตฟอร์มเกมสล็อตออนไลน์ที่ไม่ได้แค่ให้คุณเล่น แม้กระนั้นพาคุณทำเงินอย่างชาญฉลาด เว็บไซต์นี้เน้นที่การให้บริการแบบสุจริตใจ เกมที่คัดสรรมาเฉพาะตัวแตกไว ฟีพบร์ล้ำ และโบนัสแจกจริงวันแล้ววันเล่า ไม่ต้องหวังพึ่งดวงอย่างเดียวอีกต่อไป เนื่องจากระบบหลังบ้านของ สล็อต168 ถูกวางแบบมาเพื่อ “ช่วยเหลือ” ให้ผู้เล่นมีโอกาสสร้างรายได้เยอะที่สุด สล็อตเว็บไซต์ตรงชั้น 1 ที่ทำให้ท่านไม่ได้อยากต้องการกลับไปเล่นเว็บเดิมอีกเลย
สล็อตเว็บไซต์ตรงชั้น 1 ที่ทำให้ท่านไม่ได้อยากต้องการกลับไปเล่นเว็บเดิมอีกเลย

 หากคุณพร้อมเล่น
หากคุณพร้อมเล่น  เริ่มต้นด้วยเบทต่ำสุด: เริ่มต้นเดิมพันด้วยเบทขั้นต่ำสุด บางทีอาจจะ 1-2 บาทก็ได้ เพื่อคุณได้จับจังหวะและมองว่าเกมกำลังเข้าสู่ไลน์เดิมพันที่ชนะมากขึ้นเรื่อยๆ หรือกำลังแจกโบนัสอยู่ไหม เป็นการประเมินเกม
เริ่มต้นด้วยเบทต่ำสุด: เริ่มต้นเดิมพันด้วยเบทขั้นต่ำสุด บางทีอาจจะ 1-2 บาทก็ได้ เพื่อคุณได้จับจังหวะและมองว่าเกมกำลังเข้าสู่ไลน์เดิมพันที่ชนะมากขึ้นเรื่อยๆ หรือกำลังแจกโบนัสอยู่ไหม เป็นการประเมินเกม
 บาคาร่า ได้รับความไว้วางใจจากผู้ใช้งานเยอะมากๆ ไม่เพียงแค่เนื่องจากการมีระบบที่ล้ำสมัยและก็ไม่เป็นอันตราย แม้กระนั้นยังเนื่องมาจากการให้บริการที่มีคุณภาพ แล้วก็การโต้ตอบต่อความอยากของผู้เล่นที่เยี่ยมที่สุด ซึ่งความเชื่อมั่นนี้ชี้ให้เห็นว่าผู้เล่นที่เลือกเล่นกับ Sexyauto168 สามารถได้รับประสบการณ์ที่ปลอดภัยรวมทั้งเพลิดเพลินใจไปกับการเล่นบาคาร่าออนไลน์ได้อย่างมาก
บาคาร่า ได้รับความไว้วางใจจากผู้ใช้งานเยอะมากๆ ไม่เพียงแค่เนื่องจากการมีระบบที่ล้ำสมัยและก็ไม่เป็นอันตราย แม้กระนั้นยังเนื่องมาจากการให้บริการที่มีคุณภาพ แล้วก็การโต้ตอบต่อความอยากของผู้เล่นที่เยี่ยมที่สุด ซึ่งความเชื่อมั่นนี้ชี้ให้เห็นว่าผู้เล่นที่เลือกเล่นกับ Sexyauto168 สามารถได้รับประสบการณ์ที่ปลอดภัยรวมทั้งเพลิดเพลินใจไปกับการเล่นบาคาร่าออนไลน์ได้อย่างมาก บาคาร่า168 เครดิตฟรี เล่นเสียเราคืนเงินให้ โปรเด็ดเฉพาะสมาชิกใหม่
บาคาร่า168 เครดิตฟรี เล่นเสียเราคืนเงินให้ โปรเด็ดเฉพาะสมาชิกใหม่

 โปรโมชั่นสล็อตเครดิตฟรี 100 ไม่ต้องฝาก
โปรโมชั่นสล็อตเครดิตฟรี 100 ไม่ต้องฝาก

 สล็อต888 ซึ่งเป็นเว็บไซต์ที่จะนำพาความมั่งคั่งความสำเร็จและก็สิ่งดีๆให้ท่านได้รับเป็นการทดแทนเพื่อทำให้สมาชิกได้รับเงินรางวัลอย่างสุขสบายที่สุด อีกทั้งยังเป็นเหตุให้คุณได้รับความสามารถในการเล่นเกมเสี่ยงโชคออนไลน์ที่มีมาตรฐาน ซึ่งเว็บไซต์แห่งนี้พวกเราขอรับรองเลยว่าเป็นเว็บไซต์ที่จะสร้างความมั่นคงและยั่งยืนสุจริตใจวางใจได้ให้กับคุณ รวมทั้งที่สำคัญคือคุณยังมีความพร้อมเพรียงในการเล่นเกมอย่างมีประสิทธิภาพ วันนี้เราจะพาคุณไปดูกันว่าวิธีสำหรับการเลือกเว็บไซต์สล็อตที่สร้างความรวยให้กับคุณมีอะไรบ้าง
สล็อต888 ซึ่งเป็นเว็บไซต์ที่จะนำพาความมั่งคั่งความสำเร็จและก็สิ่งดีๆให้ท่านได้รับเป็นการทดแทนเพื่อทำให้สมาชิกได้รับเงินรางวัลอย่างสุขสบายที่สุด อีกทั้งยังเป็นเหตุให้คุณได้รับความสามารถในการเล่นเกมเสี่ยงโชคออนไลน์ที่มีมาตรฐาน ซึ่งเว็บไซต์แห่งนี้พวกเราขอรับรองเลยว่าเป็นเว็บไซต์ที่จะสร้างความมั่นคงและยั่งยืนสุจริตใจวางใจได้ให้กับคุณ รวมทั้งที่สำคัญคือคุณยังมีความพร้อมเพรียงในการเล่นเกมอย่างมีประสิทธิภาพ วันนี้เราจะพาคุณไปดูกันว่าวิธีสำหรับการเลือกเว็บไซต์สล็อตที่สร้างความรวยให้กับคุณมีอะไรบ้าง





 เข้ามาเล่นสล็อตเครดิตฟรีทั้งที เราก็จำเป็นต้องตั้งอกตั้งใจสำหรับเพื่อการทำกำไรจากการเล่นpanda555ให้เพิ่มมากขึ้นหน่อยสิขอรับจริงไหม? การเล่นPanda555เป็นช่องทางที่ดีที่จะทำให้คุณสามารถทำเงินได้ แล้วก็ยิ่งมี
เข้ามาเล่นสล็อตเครดิตฟรีทั้งที เราก็จำเป็นต้องตั้งอกตั้งใจสำหรับเพื่อการทำกำไรจากการเล่นpanda555ให้เพิ่มมากขึ้นหน่อยสิขอรับจริงไหม? การเล่นPanda555เป็นช่องทางที่ดีที่จะทำให้คุณสามารถทำเงินได้ แล้วก็ยิ่งมี • เล่นในขณะที่แจ็คพอตแตกง่าย: การเลือกช่วงเวลาสำหรับการเล่นสล็อตเครดิตฟรีก็มีผลต่อแนวทางการทำกำไรด้วยเหมือนกันครับ โดยเฉพาะอย่างยิ่งช่วงที่คนเล่นน้อยๆแจ็คพอตชอบแตกง่ายดายมากยิ่งขึ้น เหมาะกับการเล่นแพนด้า555เพื่อทำกำไรอย่างแท้จริงเลยขอรับ
• เล่นในขณะที่แจ็คพอตแตกง่าย: การเลือกช่วงเวลาสำหรับการเล่นสล็อตเครดิตฟรีก็มีผลต่อแนวทางการทำกำไรด้วยเหมือนกันครับ โดยเฉพาะอย่างยิ่งช่วงที่คนเล่นน้อยๆแจ็คพอตชอบแตกง่ายดายมากยิ่งขึ้น เหมาะกับการเล่นแพนด้า555เพื่อทำกำไรอย่างแท้จริงเลยขอรับ

 สล็อต888เว็บตรงกลายเป็นตัวเลือกที่นักพนันคนไม่ใช่น้อยมีความสนใจ ด้วยความสะดวกสบายและโอกาสสำหรับเพื่อการทำเงินที่มีมากขึ้น วันนี้เราจะพาทุกคนไปรู้จักกับ jinda888 อันดับ 1 ที่มีการค้ำประกันว่าเป็นเว็บแท้ 100% รวมทั้งรับรองว่าเล่นแล้วสามารถทำกำไรได้จริง จากการแตกง่ายและก็แตกหนักของเกมสล็อต888จากค่ายใหญ่ที่ได้รับการยินยอมรับทั้งโลก
สล็อต888เว็บตรงกลายเป็นตัวเลือกที่นักพนันคนไม่ใช่น้อยมีความสนใจ ด้วยความสะดวกสบายและโอกาสสำหรับเพื่อการทำเงินที่มีมากขึ้น วันนี้เราจะพาทุกคนไปรู้จักกับ jinda888 อันดับ 1 ที่มีการค้ำประกันว่าเป็นเว็บแท้ 100% รวมทั้งรับรองว่าเล่นแล้วสามารถทำกำไรได้จริง จากการแตกง่ายและก็แตกหนักของเกมสล็อต888จากค่ายใหญ่ที่ได้รับการยินยอมรับทั้งโลก เว็บไซต์สล็อตนั่นเป็นเว็บไซต์ที่เปิดให้บริการเกมjinda888โดยไม่ผ่านเอเย่นต์ ซึ่งทำให้ผู้เล่นได้รับผลประโยชน์เต็มๆโดยไม่มีการหักค่าธรรมเนียมหรือค่าคอมไม่ชชันอะไรก็ตามโดยตรงจากผู้ให้บริการเกมjinda888จากค่ายใหญ่ต่างๆยกตัวอย่างเช่น PG Slot,
เว็บไซต์สล็อตนั่นเป็นเว็บไซต์ที่เปิดให้บริการเกมjinda888โดยไม่ผ่านเอเย่นต์ ซึ่งทำให้ผู้เล่นได้รับผลประโยชน์เต็มๆโดยไม่มีการหักค่าธรรมเนียมหรือค่าคอมไม่ชชันอะไรก็ตามโดยตรงจากผู้ให้บริการเกมjinda888จากค่ายใหญ่ต่างๆยกตัวอย่างเช่น PG Slot, 
 มาทำความรู้จักกับ เว็บสล็อต กันสักนิดดีมากยิ่งกว่า! พวกเราเป็นสล็อตเว็บตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง ลิขสิทธิ์แท้จากค่ายเกมชั้นนำระดับนานาชาติล้นหลาม มาพร้อมเกมlive22แบบจัดเต็มกว่า 2,000 เกม สามารถเล่นผ่านโปรแกรมสำหรับเล่นอินเตอร์เน็ตได้ในทันที ไม่ต้องดาวน์โหลดหรือจัดตั้ง สมัครแล้วรับโปรโมชั่นแบบจัดเต็มได้เลย บอกเลยว่า คนใดที่เข้ามาเล่นเกมกับเรา เว็บสล็อต ในเวลานี้จำเป็นที่จะต้องชอบใจแน่ๆ เนื่องจากพวกเรามีเกมเก่าแล้วก็ใหม่มากมาย รวมเกมสล็อตแตกง่ายแตกจริงจากค่ายเกมต่างๆอีกมากมาย สมัครสมาชิกใหม่ live22slotฝากถอนง่ายไม่มีอย่างน้อย ใช้งานง่ายทุกระบบแบบ 100% อีกด้วย ลงทะเบียนสมัครสมาชิกฟรี สมัครเลย!
มาทำความรู้จักกับ เว็บสล็อต กันสักนิดดีมากยิ่งกว่า! พวกเราเป็นสล็อตเว็บตรงไม่ผ่านเอเย่นต์ ไม่ผ่านตัวกลาง ลิขสิทธิ์แท้จากค่ายเกมชั้นนำระดับนานาชาติล้นหลาม มาพร้อมเกมlive22แบบจัดเต็มกว่า 2,000 เกม สามารถเล่นผ่านโปรแกรมสำหรับเล่นอินเตอร์เน็ตได้ในทันที ไม่ต้องดาวน์โหลดหรือจัดตั้ง สมัครแล้วรับโปรโมชั่นแบบจัดเต็มได้เลย บอกเลยว่า คนใดที่เข้ามาเล่นเกมกับเรา เว็บสล็อต ในเวลานี้จำเป็นที่จะต้องชอบใจแน่ๆ เนื่องจากพวกเรามีเกมเก่าแล้วก็ใหม่มากมาย รวมเกมสล็อตแตกง่ายแตกจริงจากค่ายเกมต่างๆอีกมากมาย สมัครสมาชิกใหม่ live22slotฝากถอนง่ายไม่มีอย่างน้อย ใช้งานง่ายทุกระบบแบบ 100% อีกด้วย ลงทะเบียนสมัครสมาชิกฟรี สมัครเลย! • ระบบฝากถอนออโต้:
• ระบบฝากถอนออโต้:  • ฟีพบร์ช่วยเหลือเยอะมาก: หากคุณเข้ามาใช้บริการLive22กับเราเว็บตรง คุณจะพบว่า บนเว็บเว็บสล็อตของเรามีช่องทางสำหรับเพื่อการช่วยเหลือผู้เล่นใหม่มากไม่น้อยเลยทีเดียว ไม่ว่าจะเป็นบทความแนะนำการเล่นหรือชี้แนะเกมต่างๆรวมถึงช่องทางสำหรับเพื่อการติดต่อกับทีมงานได้ตลอดเวลาอีกด้วย
• ฟีพบร์ช่วยเหลือเยอะมาก: หากคุณเข้ามาใช้บริการLive22กับเราเว็บตรง คุณจะพบว่า บนเว็บเว็บสล็อตของเรามีช่องทางสำหรับเพื่อการช่วยเหลือผู้เล่นใหม่มากไม่น้อยเลยทีเดียว ไม่ว่าจะเป็นบทความแนะนำการเล่นหรือชี้แนะเกมต่างๆรวมถึงช่องทางสำหรับเพื่อการติดต่อกับทีมงานได้ตลอดเวลาอีกด้วย ลงทะเบียนกับพวกเราผ่าน ปาก
ลงทะเบียนกับพวกเราผ่าน ปาก มองหา
มองหา  5 สัญญาณที่บอกว่า คุณควรหยุดเล่นบาคาร่าได้แล้ว!
5 สัญญาณที่บอกว่า คุณควรหยุดเล่นบาคาร่าได้แล้ว! • แพ้ต่อเนื่องกันหลายตา: ถ้าหากคุณเริ่มแพ้ต่อเนื่องกันมากยิ่งกว่า 5-7 ตา ถือว่าเป็นสัญญาณที่ไม่ดีแล้วครับ คุณควรจะหยุดเล่นในทันที ยิ่งหากคุณเล่นแพ้แล้วแทงทบเมื่อแพ้ด้วยแล้วล่ะก็ คุณคงจะหมดทุนไปเยอะๆแน่นอน ด้วยเหตุนี้ ควรพักสัก 10-15 นาที หรือจะหยุดเล่นในวันนั้นไปเลยดีแล้วครับผม ด้วยเหตุว่าน่าจะไม่ใช่จังหวะสำหรับในการทำกำไรที่ดีของคุณแน่นอน
• แพ้ต่อเนื่องกันหลายตา: ถ้าหากคุณเริ่มแพ้ต่อเนื่องกันมากยิ่งกว่า 5-7 ตา ถือว่าเป็นสัญญาณที่ไม่ดีแล้วครับ คุณควรจะหยุดเล่นในทันที ยิ่งหากคุณเล่นแพ้แล้วแทงทบเมื่อแพ้ด้วยแล้วล่ะก็ คุณคงจะหมดทุนไปเยอะๆแน่นอน ด้วยเหตุนี้ ควรพักสัก 10-15 นาที หรือจะหยุดเล่นในวันนั้นไปเลยดีแล้วครับผม ด้วยเหตุว่าน่าจะไม่ใช่จังหวะสำหรับในการทำกำไรที่ดีของคุณแน่นอน • เลือกห้องแล้วก็สังเกตดีลเลอร์: ในตอนที่คุณได้ห้องพนันแล้ว ให้ท่านสังเกตดีลเลอร์ก่อนว่า พวกเขาออกไพ่ บาคาร่า168 แบบไหนหลายครั้งที่สุด เพื่อที่คุณจะได้เลือกแทงตามแนวทางที่คุณมองเห็นเป็นประจำจากดีลเลอร์คนนั้นนั่นเองครับผม
• เลือกห้องแล้วก็สังเกตดีลเลอร์: ในตอนที่คุณได้ห้องพนันแล้ว ให้ท่านสังเกตดีลเลอร์ก่อนว่า พวกเขาออกไพ่ บาคาร่า168 แบบไหนหลายครั้งที่สุด เพื่อที่คุณจะได้เลือกแทงตามแนวทางที่คุณมองเห็นเป็นประจำจากดีลเลอร์คนนั้นนั่นเองครับผม • ความปลอดภัยสูง
• ความปลอดภัยสูง สำหรับผม การได้ทดลองเล่นเกมสล็อตใหม่ๆที่พึ่งจะอัปเดตจาก ทางเข้าpg เป็นประสบการณ์ที่น่าตื่นตาตื่นใจและคุ้มค่ามากมาย ด้วยเหตุว่าแต่ละเกมทางเข้าpgได้รับการออกแบบมาอย่างพิถีพิถันเพื่อให้ผู้เล่นทุกคนมีโอกาสสำหรับเพื่อการได้กำไรได้จริง เกมfunny18ใหม่แต่ละเกมมิได้มีแค่กราฟิกที่งดงาม แต่ว่ายังมาพร้อมกับฟีพบร์พิเศษที่ช่วยทำให้การลุ้นแจ็คพอตเป็นเรื่องที่ไม่ได้ยากเย็นขึ้นกว่าเดิม
สำหรับผม การได้ทดลองเล่นเกมสล็อตใหม่ๆที่พึ่งจะอัปเดตจาก ทางเข้าpg เป็นประสบการณ์ที่น่าตื่นตาตื่นใจและคุ้มค่ามากมาย ด้วยเหตุว่าแต่ละเกมทางเข้าpgได้รับการออกแบบมาอย่างพิถีพิถันเพื่อให้ผู้เล่นทุกคนมีโอกาสสำหรับเพื่อการได้กำไรได้จริง เกมfunny18ใหม่แต่ละเกมมิได้มีแค่กราฟิกที่งดงาม แต่ว่ายังมาพร้อมกับฟีพบร์พิเศษที่ช่วยทำให้การลุ้นแจ็คพอตเป็นเรื่องที่ไม่ได้ยากเย็นขึ้นกว่าเดิม • ฝาก 300 บาท รับฟรี 100 บาท
• ฝาก 300 บาท รับฟรี 100 บาท อีสปอร์ตหรือกีฬาอิเล็กทรอนิกส์ได้รับความนิยมอย่างมากในทศวรรษที่ผ่านมา โดยเฉพาะในยุคดิจิทัลที่มีการพัฒนาเทคโนโลยีและอินเทอร์เน็ตที่สามารถเชื่อมต่อผู้คนจากทั่วทุกมุมโลกได้อย่างง่ายดาย อีสปอร์ตไม่เพียงแต่เป็นการแข่งขันวิดีโอเกมระดับมืออาชีพเท่านั้น แต่ยังได้รับการยอมรับว่าเป็นกีฬาชนิดหนึ่งที่มีการจัดแข่งขันอย่างเป็นทางการ โดยมีผู้ชมและแฟนคลับเพิ่มขึ้นอย่างต่อเนื่อง
อีสปอร์ตหรือกีฬาอิเล็กทรอนิกส์ได้รับความนิยมอย่างมากในทศวรรษที่ผ่านมา โดยเฉพาะในยุคดิจิทัลที่มีการพัฒนาเทคโนโลยีและอินเทอร์เน็ตที่สามารถเชื่อมต่อผู้คนจากทั่วทุกมุมโลกได้อย่างง่ายดาย อีสปอร์ตไม่เพียงแต่เป็นการแข่งขันวิดีโอเกมระดับมืออาชีพเท่านั้น แต่ยังได้รับการยอมรับว่าเป็นกีฬาชนิดหนึ่งที่มีการจัดแข่งขันอย่างเป็นทางการ โดยมีผู้ชมและแฟนคลับเพิ่มขึ้นอย่างต่อเนื่อง pgslot เว็บตรงไม่ผ่านเอเย่นต์ แตกง่าย ฝากถอนออโต้ สมัครฟรี
pgslot เว็บตรงไม่ผ่านเอเย่นต์ แตกง่าย ฝากถอนออโต้ สมัครฟรี 1. ความปลอดภัยแล้วก็ความน่าไว้วางใจ
1. ความปลอดภัยแล้วก็ความน่าไว้วางใจ
 เกมสล็อตนานัปการที่ โจ๊กเกอร์123 ความน่านับถือแล้วก็ความปลอดภัย
เกมสล็อตนานัปการที่ โจ๊กเกอร์123 ความน่านับถือแล้วก็ความปลอดภัย โอกาสที่สบายและตอบโจทย์ผู้เล่นjoker123ได้เป็นอย่างดี แล้วก็ สล็อตโจ๊กเกอร์ คือหนึ่งในตัวเลือกที่ไม่ควรพลาด! มาพร้อมกับการเล่นที่ไม่ต้องดาวน์โหลดแอปพลิเคชัน เพิ่มความสะดวกสบายสำหรับเพื่อการเล่นเกมและก็เข้าถึงเกมได้ทุกที่ที่ต้องการ เพียงมีมือถือและก็อินเทอร์เน็ตเท่านั้น
โอกาสที่สบายและตอบโจทย์ผู้เล่นjoker123ได้เป็นอย่างดี แล้วก็ สล็อตโจ๊กเกอร์ คือหนึ่งในตัวเลือกที่ไม่ควรพลาด! มาพร้อมกับการเล่นที่ไม่ต้องดาวน์โหลดแอปพลิเคชัน เพิ่มความสะดวกสบายสำหรับเพื่อการเล่นเกมและก็เข้าถึงเกมได้ทุกที่ที่ต้องการ เพียงมีมือถือและก็อินเทอร์เน็ตเท่านั้น
 ประวัติความเป็นมาและก็ที่มาที่ไป
ประวัติความเป็นมาและก็ที่มาที่ไป เว้นเสียแต่เกมรากฐานแล้ว macau888 ยังมีการอัปเดตเกมใหม่ๆอยู่เป็นประจำ ทำให้ผู้เล่นไม่ทราบสึกเบื่อแล้วก็สามารถทดลองเล่นเกมใหม่ๆได้อย่างต่อเนื่อง
เว้นเสียแต่เกมรากฐานแล้ว macau888 ยังมีการอัปเดตเกมใหม่ๆอยู่เป็นประจำ ทำให้ผู้เล่นไม่ทราบสึกเบื่อแล้วก็สามารถทดลองเล่นเกมใหม่ๆได้อย่างต่อเนื่อง โปรแกรมสะสมแต้มช่วยให้ผู้เล่นได้รับคุณประโยชน์เพิ่มขึ้นในทุกการเดิมพัน โดยสามารถแลกของรางวัลหรือเครดิตเดิมพันเสริมเติมได้
โปรแกรมสะสมแต้มช่วยให้ผู้เล่นได้รับคุณประโยชน์เพิ่มขึ้นในทุกการเดิมพัน โดยสามารถแลกของรางวัลหรือเครดิตเดิมพันเสริมเติมได้ 5. ประสบการณ์แล้วก็รีวิวจากผู้เล่นจริง
5. ประสบการณ์แล้วก็รีวิวจากผู้เล่นจริง สวัสดีขอรับ ผมเป็นคนนึงที่ถูกใจเล่น
สวัสดีขอรับ ผมเป็นคนนึงที่ถูกใจเล่น


 ทำไมบริการทดสอบเล่นสล็อตฟรีถึงสำคัญ?
ทำไมบริการทดสอบเล่นสล็อตฟรีถึงสำคัญ? ผมมองว่าการทดลองเล่นTangtem168ฟรีคือจังหวะที่สามารถจะช่วยให้เราได้ทำความเข้าใจเกมสล็อตอย่างลึกซึ้ง อีกทั้งในเรื่องของแบบอย่างเกม ฟีเจอร์พิเศษต่างๆแล้วก็แนวทางการเล่นที่สมควร สำหรับมือใหม่ การได้ลองเล่นเกมก่อนช่วยให้เข้าใจกฎกติการวมทั้งความเพลิดเพลินของเกมนั้นๆโดยไม่ต้องเสียเงินเสียทองเลย
ผมมองว่าการทดลองเล่นTangtem168ฟรีคือจังหวะที่สามารถจะช่วยให้เราได้ทำความเข้าใจเกมสล็อตอย่างลึกซึ้ง อีกทั้งในเรื่องของแบบอย่างเกม ฟีเจอร์พิเศษต่างๆแล้วก็แนวทางการเล่นที่สมควร สำหรับมือใหม่ การได้ลองเล่นเกมก่อนช่วยให้เข้าใจกฎกติการวมทั้งความเพลิดเพลินของเกมนั้นๆโดยไม่ต้องเสียเงินเสียทองเลย กระบวนการลงทะเบียนรับเครดิตฟรี
กระบวนการลงทะเบียนรับเครดิตฟรี
 สำหรับบทความนี้ ผมจะพาทุกคนไปรู้จักกับ พุซซี่888 ให้มากขึ้นเรื่อยๆ ทั้งยังเรื่องระบบการใช้งาน โปรโมชั่นพิเศษ แล้วก็แนวทางการลงทะเบียนเป็นสมาชิก รวมทั้งเหตุผลว่าเพราะเหตุไรเว็บไซต์แห่งนี้ถึงเป็นตัวเลือกที่เหมาะสมที่สุดสำหรับนักปั่นสล็อตในปี 2024 ถ้าเกิดคุณกำลังมองหาเว็บไซต์ที่แตกง่าย จ่ายจริง รวมทั้งมีความน่านับถือ ห้ามพลาดบทความนี้ครับ
สำหรับบทความนี้ ผมจะพาทุกคนไปรู้จักกับ พุซซี่888 ให้มากขึ้นเรื่อยๆ ทั้งยังเรื่องระบบการใช้งาน โปรโมชั่นพิเศษ แล้วก็แนวทางการลงทะเบียนเป็นสมาชิก รวมทั้งเหตุผลว่าเพราะเหตุไรเว็บไซต์แห่งนี้ถึงเป็นตัวเลือกที่เหมาะสมที่สุดสำหรับนักปั่นสล็อตในปี 2024 ถ้าเกิดคุณกำลังมองหาเว็บไซต์ที่แตกง่าย จ่ายจริง รวมทั้งมีความน่านับถือ ห้ามพลาดบทความนี้ครับ

 เนื้อหานี้ผมจะมารีทิวทัศน์แบบละเอียดว่า สล็อตเว็บตรง ดีอย่างไร? เพราะอะไรถึงเป็นเว็บสล็อตที่คุณไม่สมควรพลาด พร้อมชี้แนะเคล็ดลับเล่นสล็อตให้ได้เงิน รวมทั้งเนื้อหาเกี่ยวกับ โปรโมชั่นเด็ดๆที่คุณควรจะรู้ ถ้าเกิดพร้อมแล้ว มาดูกันเลยขอรับ!
เนื้อหานี้ผมจะมารีทิวทัศน์แบบละเอียดว่า สล็อตเว็บตรง ดีอย่างไร? เพราะอะไรถึงเป็นเว็บสล็อตที่คุณไม่สมควรพลาด พร้อมชี้แนะเคล็ดลับเล่นสล็อตให้ได้เงิน รวมทั้งเนื้อหาเกี่ยวกับ โปรโมชั่นเด็ดๆที่คุณควรจะรู้ ถ้าเกิดพร้อมแล้ว มาดูกันเลยขอรับ! ข้อตกลงการรับเครดิตฟรี
ข้อตกลงการรับเครดิตฟรี นอกเหนือจากความมากมายหลายของเกมแล้ว gaojing888 ยังมาพร้อมกับระบบฝากถอนออโต้ที่เร็ว ไม่ต้องรอนาน รองรับทั้งยังการฝากผ่านแบงค์และก็ True Wallet ทำให้สบายต่อการใช้งาน ทั้งยังมีโปรโมชั่นสุดพิเศษ เป็นต้นว่า สมัครเป็นสมาชิกใหม่รับเครดิตฟรี ไม่ต้องฝาก ไม่ต้องแชร์ ตอบปัญหาผู้เล่นทุกระดับทั้งมือใหม่แล้วก็มือโปร
นอกเหนือจากความมากมายหลายของเกมแล้ว gaojing888 ยังมาพร้อมกับระบบฝากถอนออโต้ที่เร็ว ไม่ต้องรอนาน รองรับทั้งยังการฝากผ่านแบงค์และก็ True Wallet ทำให้สบายต่อการใช้งาน ทั้งยังมีโปรโมชั่นสุดพิเศษ เป็นต้นว่า สมัครเป็นสมาชิกใหม่รับเครดิตฟรี ไม่ต้องฝาก ไม่ต้องแชร์ ตอบปัญหาผู้เล่นทุกระดับทั้งมือใหม่แล้วก็มือโปร

 สำหรับคนไหนกันแน่ที่ยังลังเลว่าจะเลือกเล่นบาคาร่ากับเว็บไหนดี บาคาร่า มีจุดแข็งที่น่าสนใจหลายชนิด ไม่ว่าจะเป็นระบบฝาก-ถอนอัตโนมัติที่รวดเร็วทันใจ โปรโมชั่นคืนเงินยอดเสีย และยังรวมไปถึงบริการทดสอบเล่นบาคาร่าฟรี ซึ่งทั้งสิ้นนี้จะช่วยทำให้คุณแน่ใจว่า การเดิมพันของคุณจะเป็นไปอย่างราบรื่นรวมทั้งครึกครื้น ถ้าหากพร้อมแล้ว มาเริ่มดูกันเลยนะครับว่า sexyauto168 มีอะไรที่น่าดึงดูดบ้าง
สำหรับคนไหนกันแน่ที่ยังลังเลว่าจะเลือกเล่นบาคาร่ากับเว็บไหนดี บาคาร่า มีจุดแข็งที่น่าสนใจหลายชนิด ไม่ว่าจะเป็นระบบฝาก-ถอนอัตโนมัติที่รวดเร็วทันใจ โปรโมชั่นคืนเงินยอดเสีย และยังรวมไปถึงบริการทดสอบเล่นบาคาร่าฟรี ซึ่งทั้งสิ้นนี้จะช่วยทำให้คุณแน่ใจว่า การเดิมพันของคุณจะเป็นไปอย่างราบรื่นรวมทั้งครึกครื้น ถ้าหากพร้อมแล้ว มาเริ่มดูกันเลยนะครับว่า sexyauto168 มีอะไรที่น่าดึงดูดบ้าง
 สล็อตเว็บตรง อันดับ 1 ในไทย ไม่มีประวัติการทุจริต ไม่มีล็อคผลแน่ๆ!
สล็อตเว็บตรง อันดับ 1 ในไทย ไม่มีประวัติการทุจริต ไม่มีล็อคผลแน่ๆ!

 สล็อต แบบครบวงจรศูนย์รวมเกมยอดฮิต พร้อมสนุกสนานไปกับเว็บ Jinda55 สล็อตที่ดีที่สุด การเล่นเกมคาสิโนออนไลน์เปลี่ยนเป็นหนึ่งในความบันเทิงยอดฮิต การค้นหาเว็บไซต์ที่ตอบโจทย์ทุกความต้องการเปลี่ยนเป็นสิ่งสำคัญอย่างมาก เราจะพาคุณไปรู้จักกับบริการคาสิโนออนไลน์แบบครบวงจรที่รวบรวมเกมยอดฮิตเอาไว้ภายในที่เดียว พร้อมเสนอแนะเว็บสล็อตที่เหมาะสมที่สุดที่ให้ประสบการณ์การเล่นที่น่าเร้าใจและก็คุ้มค่าที่สุด
สล็อต แบบครบวงจรศูนย์รวมเกมยอดฮิต พร้อมสนุกสนานไปกับเว็บ Jinda55 สล็อตที่ดีที่สุด การเล่นเกมคาสิโนออนไลน์เปลี่ยนเป็นหนึ่งในความบันเทิงยอดฮิต การค้นหาเว็บไซต์ที่ตอบโจทย์ทุกความต้องการเปลี่ยนเป็นสิ่งสำคัญอย่างมาก เราจะพาคุณไปรู้จักกับบริการคาสิโนออนไลน์แบบครบวงจรที่รวบรวมเกมยอดฮิตเอาไว้ภายในที่เดียว พร้อมเสนอแนะเว็บสล็อตที่เหมาะสมที่สุดที่ให้ประสบการณ์การเล่นที่น่าเร้าใจและก็คุ้มค่าที่สุด แพลตฟอร์มที่รวบรวมเกม Jinda55 หลากหลายแบบอย่างไว้ในที่เดียว ไม่ว่าจะเป็นสล็อต บาคาร่า รูเล็ต ไฮโล โป๊กเกอร์ หรือแม้แต่เกมยิงปลา คุณสามารถเล่นทุกสิ่งทุกอย่างได้ภายใต้บัญชีผู้ใช้งานเดียว สะดวกและก็เหมาะกับผู้เล่นที่ปรารถนาสัมผัสประสบการณ์มากมายแบบไม่ต้องย้ายแพลตฟอร์มเลย
แพลตฟอร์มที่รวบรวมเกม Jinda55 หลากหลายแบบอย่างไว้ในที่เดียว ไม่ว่าจะเป็นสล็อต บาคาร่า รูเล็ต ไฮโล โป๊กเกอร์ หรือแม้แต่เกมยิงปลา คุณสามารถเล่นทุกสิ่งทุกอย่างได้ภายใต้บัญชีผู้ใช้งานเดียว สะดวกและก็เหมาะกับผู้เล่นที่ปรารถนาสัมผัสประสบการณ์มากมายแบบไม่ต้องย้ายแพลตฟอร์มเลย

 สิ่งที่ทำให้ joker123ths ไม่เหมือนกันรวมทั้งเด่นกว่าเว็บไซต์อื่นๆเป็นการเป็นเว็บตรงไม่ผ่านเอเย่นต์ และก็ระบบเดิมพันไม่มีอย่างต่ำ ซึ่งเหมาะกับทุกคน ไม่ว่าคุณจะมีทุนมากมายหรือน้อย ที่นี่มีเกมสล็อตจากค่ายดังให้เลือกเล่นกว่า 20 ค่าย และก็โปรโมชั่นที่จำเป็นต้องพูดว่า “คุ้มค่าเกินคำพรรณนา” ถ้าคุณกำลังมองหาเว็บที่น่าไว้วางใจ ไม่เป็นอันตราย และเต็มไปด้วยโอกาสทำเงิน joker123thเป็นตัวเลือกอันดับต้นๆของผม
สิ่งที่ทำให้ joker123ths ไม่เหมือนกันรวมทั้งเด่นกว่าเว็บไซต์อื่นๆเป็นการเป็นเว็บตรงไม่ผ่านเอเย่นต์ และก็ระบบเดิมพันไม่มีอย่างต่ำ ซึ่งเหมาะกับทุกคน ไม่ว่าคุณจะมีทุนมากมายหรือน้อย ที่นี่มีเกมสล็อตจากค่ายดังให้เลือกเล่นกว่า 20 ค่าย และก็โปรโมชั่นที่จำเป็นต้องพูดว่า “คุ้มค่าเกินคำพรรณนา” ถ้าคุณกำลังมองหาเว็บที่น่าไว้วางใจ ไม่เป็นอันตราย และเต็มไปด้วยโอกาสทำเงิน joker123thเป็นตัวเลือกอันดับต้นๆของผม 1. ความเร็วสำหรับเพื่อการทำธุรกรรม
1. ความเร็วสำหรับเพื่อการทำธุรกรรม
 สล็อตเว็บไซต์ตรง ลงทะเบียนสมัครสมาชิกรับเครดิตฟรี พร้อมบริการทดสอบเล่นสล็อตฟรี ไม่ต้องจ่ายเงิน
สล็อตเว็บไซต์ตรง ลงทะเบียนสมัครสมาชิกรับเครดิตฟรี พร้อมบริการทดสอบเล่นสล็อตฟรี ไม่ต้องจ่ายเงิน บทความนี้ ผมตั้งใจเขียนให้ทุกคนที่กำลังสนใจเริ่มต้นเล่นสล็อต หรือแม้แต่คนที่เล่นสล็อตเว็บตรงมานานแต่ว่ายังหาเว็บไซต์ที่ใช่ ไม่พบ ผมจะพาไปดูทุกมุมมองของ สล็อต99 ไม่ว่าจะเป็นลักษณะเด่น ระบบทดสอบเล่นฟรี และแนวทางการเล่น
บทความนี้ ผมตั้งใจเขียนให้ทุกคนที่กำลังสนใจเริ่มต้นเล่นสล็อต หรือแม้แต่คนที่เล่นสล็อตเว็บตรงมานานแต่ว่ายังหาเว็บไซต์ที่ใช่ ไม่พบ ผมจะพาไปดูทุกมุมมองของ สล็อต99 ไม่ว่าจะเป็นลักษณะเด่น ระบบทดสอบเล่นฟรี และแนวทางการเล่น
 การเล่น สล็อต168 เป็นที่นิยมอย่างล้นหลามในขณะนี้ ด้วยความสบายสบายสำหรับการเล่นผ่านระบบออนไลน์ และจังหวะสำหรับในการได้กำไรที่ไม่ยากเกินความจำเป็น ผู้คนจำนวนไม่ใช้น้อยที่ชื่นชอบเกี่ยวกับการเสี่ยงดวงจึงหันมาพอใจเกมสล็อตออนไลน์เยอะขึ้น โดยยิ่งไปกว่านั้นเกมสล็อตที่ให้บริการผ่านเว็บไซต์ตรงหรือเว็บสล็อตแท้ ซึ่งมีความน่าไว้วางใจและปลอดภัย วันนี้พวกเราจะพาคุณไปทำความรู้จักกับ สล็อตเว็บไซต์ตรง ซึ่งยอดเยี่ยมในค่ายดังยอดนิยมที่นักพนันสนใจสูง เนื่องจากมีการให้บริการที่ครบวงจร พร้อมช่องทางสำหรับในการแตกแจ็คพอตที่สูง
การเล่น สล็อต168 เป็นที่นิยมอย่างล้นหลามในขณะนี้ ด้วยความสบายสบายสำหรับการเล่นผ่านระบบออนไลน์ และจังหวะสำหรับในการได้กำไรที่ไม่ยากเกินความจำเป็น ผู้คนจำนวนไม่ใช้น้อยที่ชื่นชอบเกี่ยวกับการเสี่ยงดวงจึงหันมาพอใจเกมสล็อตออนไลน์เยอะขึ้น โดยยิ่งไปกว่านั้นเกมสล็อตที่ให้บริการผ่านเว็บไซต์ตรงหรือเว็บสล็อตแท้ ซึ่งมีความน่าไว้วางใจและปลอดภัย วันนี้พวกเราจะพาคุณไปทำความรู้จักกับ สล็อตเว็บไซต์ตรง ซึ่งยอดเยี่ยมในค่ายดังยอดนิยมที่นักพนันสนใจสูง เนื่องจากมีการให้บริการที่ครบวงจร พร้อมช่องทางสำหรับในการแตกแจ็คพอตที่สูง ปั่น slot168 เป็นหนึ่งในผู้ให้บริการเกมสล็อตออนไลน์ที่โด่งดังในวงการ ด้วยเหตุว่าเป็นเว็บไซต์สล็อตแท้ที่มีระบบการให้บริการที่ครบวงจร ไม่ว่าคุณจะเป็นผู้เล่นคนใหม่หรือผู้เล่นเก่าก็สามารถเข้าร่วมบันเทิงใจกับเกมสล็อตได้อย่างง่ายๆ ด้วยระบบที่มีความเสถียรภาพและไม่เป็นอันตราย พร้อมทั้งการให้บริการที่ครอบคลุมทุกด้าน ไม่ว่าจะเป็นเรื่องของการฝากถอนเงินที่รวดเร็ว หรือโบนัสพิเศษที่มีให้ในแต่ละเกม
ปั่น slot168 เป็นหนึ่งในผู้ให้บริการเกมสล็อตออนไลน์ที่โด่งดังในวงการ ด้วยเหตุว่าเป็นเว็บไซต์สล็อตแท้ที่มีระบบการให้บริการที่ครบวงจร ไม่ว่าคุณจะเป็นผู้เล่นคนใหม่หรือผู้เล่นเก่าก็สามารถเข้าร่วมบันเทิงใจกับเกมสล็อตได้อย่างง่ายๆ ด้วยระบบที่มีความเสถียรภาพและไม่เป็นอันตราย พร้อมทั้งการให้บริการที่ครอบคลุมทุกด้าน ไม่ว่าจะเป็นเรื่องของการฝากถอนเงินที่รวดเร็ว หรือโบนัสพิเศษที่มีให้ในแต่ละเกม

 พวกเราจะพาคุณมาเล่นเกมสล็อตออนไลน์ที่ได้รับความนิยมมากขึ้นอย่างเร็ว การเลือกเล่นกับ
พวกเราจะพาคุณมาเล่นเกมสล็อตออนไลน์ที่ได้รับความนิยมมากขึ้นอย่างเร็ว การเลือกเล่นกับ  เว็บไซต์ผู้ให้บริการเกมสล็อตออนไลน์ที่ได้รับลิขสิทธิ์แท้จากค่ายเกมโดยตรง ไม่ผ่านตัวแทนหรือเอเย่นต์ ทำให้ผู้เล่นมั่นใจได้ว่าจะได้รับการบริการที่มีคุณภาพ รวมทั้งการเล่นเกมที่เที่ยงธรรม ไม่มีการปรับอัตราการชนะหรือคดโกงอะไรก็แล้วแต่
เว็บไซต์ผู้ให้บริการเกมสล็อตออนไลน์ที่ได้รับลิขสิทธิ์แท้จากค่ายเกมโดยตรง ไม่ผ่านตัวแทนหรือเอเย่นต์ ทำให้ผู้เล่นมั่นใจได้ว่าจะได้รับการบริการที่มีคุณภาพ รวมทั้งการเล่นเกมที่เที่ยงธรรม ไม่มีการปรับอัตราการชนะหรือคดโกงอะไรก็แล้วแต่

 อัปเดตจัดเต็มกับเกม faro168 ในปี 2025 ที่คุณไม่สมควรพลาด
อัปเดตจัดเต็มกับเกม faro168 ในปี 2025 ที่คุณไม่สมควรพลาด Sbobet168 มาแรง ชั้น 1 ที่ สล็อตเว็บไซต์ตรง เปิดให้บริการแล้ววันนี้ ปากทางเข้าหาง่าย เล่นผ่านมือถือได้เลย
Sbobet168 มาแรง ชั้น 1 ที่ สล็อตเว็บไซต์ตรง เปิดให้บริการแล้ววันนี้ ปากทางเข้าหาง่าย เล่นผ่านมือถือได้เลย คุณกำลังมองหาเว็บสล็อตออนไลน์ที่ล้ำยุคที่สุดในตอนนี้ และปรารถนาสัมผัสประสบการณ์การพนันที่ไม่ซ้ำใคร sbobet168 สล็อตเว็บไซต์ตรง อันดับ 1 คือคำตอบที่ดีที่สุดสำหรับคุณ! เรามีบริการครบวงจร พร้อมรองรับทุกความปรารถนาของผู้เล่น อีกทั้งความปลอดภัย ความสบาย รวมทั้งจังหวะสำหรับเพื่อการชนะรางวัลใหญ่
คุณกำลังมองหาเว็บสล็อตออนไลน์ที่ล้ำยุคที่สุดในตอนนี้ และปรารถนาสัมผัสประสบการณ์การพนันที่ไม่ซ้ำใคร sbobet168 สล็อตเว็บไซต์ตรง อันดับ 1 คือคำตอบที่ดีที่สุดสำหรับคุณ! เรามีบริการครบวงจร พร้อมรองรับทุกความปรารถนาของผู้เล่น อีกทั้งความปลอดภัย ความสบาย รวมทั้งจังหวะสำหรับเพื่อการชนะรางวัลใหญ่

 แนะนำ
แนะนำ  ฟรีสปินเข้าง่าย แค่หมุนไม่กี่ครั้งก็รับโชคใหญ่
ฟรีสปินเข้าง่าย แค่หมุนไม่กี่ครั้งก็รับโชคใหญ่
 ยุคที่ทุกคนหันมาพอใจการเล่นเกมสล็อตออนไลน์มากยิ่งขึ้น เว็บไซต์สล็อตที่เปิด 24 ชั่วโมง ไม่มีทางหยุด
ยุคที่ทุกคนหันมาพอใจการเล่นเกมสล็อตออนไลน์มากยิ่งขึ้น เว็บไซต์สล็อตที่เปิด 24 ชั่วโมง ไม่มีทางหยุด  แหล่งรวมเกม slot ออนไลน์ที่หลากหลาย ปั้นทุนหลักร้อย สู่กำไรหลักแสน
แหล่งรวมเกม slot ออนไลน์ที่หลากหลาย ปั้นทุนหลักร้อย สู่กำไรหลักแสน
 รวมเกม slot แตกง่าย ครบทุกค่ายดัง รองรับมือถือ
รวมเกม slot แตกง่าย ครบทุกค่ายดัง รองรับมือถือ ไม่ว่าคุณจะเป็นมือใหม่ที่อยากลองเริ่มด้วยเงินทุนน้อยๆหรือเป็นมืออาชีพที่อยากได้ท้ากับเกมbetwayที่สลับซับซ้อนมากเพิ่มขึ้น slot จะเป็นตัวเลือกที่สมควรสำหรับคุณ อย่ารอช้า ลองสมัครสมาชิกรวมทั้งสัมผัสประสบการณ์การเล่นslotเว็บตรง ไม่ผ่านเอเย่นต์ ไม่มีขั้นต่ำ เล่นง่าย แตกง่าย ได้แล้ววันนี้ แล้วคุณจะพบว่าโลกของเกมสล็อตออนไลน์นั้นมีชีวิตชีวาแล้วก็ความสนุกสนานร่าเริงที่รอให้ท่านศึกษาค้นพบอย่างไม่รู้จบ
ไม่ว่าคุณจะเป็นมือใหม่ที่อยากลองเริ่มด้วยเงินทุนน้อยๆหรือเป็นมืออาชีพที่อยากได้ท้ากับเกมbetwayที่สลับซับซ้อนมากเพิ่มขึ้น slot จะเป็นตัวเลือกที่สมควรสำหรับคุณ อย่ารอช้า ลองสมัครสมาชิกรวมทั้งสัมผัสประสบการณ์การเล่นslotเว็บตรง ไม่ผ่านเอเย่นต์ ไม่มีขั้นต่ำ เล่นง่าย แตกง่าย ได้แล้ววันนี้ แล้วคุณจะพบว่าโลกของเกมสล็อตออนไลน์นั้นมีชีวิตชีวาแล้วก็ความสนุกสนานร่าเริงที่รอให้ท่านศึกษาค้นพบอย่างไม่รู้จบ

 การเลือกเล่นเกมjoker123นั้น ต้นสายปลายเหตุแรกๆที่ผู้เล่นมองหาเป็นความคุ้มค่า และแน่นอนว่า โจ๊กเกอร์123 เว็บไซต์สล็อตออนไลน์ เว็บตรง นั้นได้คัดสรรเกมสล็อตที่มี RTP สูง ซึ่งหมายถึงอัตราการคืนเงินให้แก่ผู้เล่นยิ่งสูง ก็ยิ่งได้โอกาสแตกง่ายมากยิ่งกว่าเดิม โดยเกมสล็อตที่เลือกสรรจากค่าย joker123 เว็บไซต์หลักนั้น มาพร้อมระบบสุ่มรางวัลที่เที่ยงธรรม ไม่มีการปรับปรุงอัตราแตกแต่อย่างใด ผู้เล่นสามารถเชื่อมั่นได้ว่าได้โอกาสชนะอย่างแท้จริง
การเลือกเล่นเกมjoker123นั้น ต้นสายปลายเหตุแรกๆที่ผู้เล่นมองหาเป็นความคุ้มค่า และแน่นอนว่า โจ๊กเกอร์123 เว็บไซต์สล็อตออนไลน์ เว็บตรง นั้นได้คัดสรรเกมสล็อตที่มี RTP สูง ซึ่งหมายถึงอัตราการคืนเงินให้แก่ผู้เล่นยิ่งสูง ก็ยิ่งได้โอกาสแตกง่ายมากยิ่งกว่าเดิม โดยเกมสล็อตที่เลือกสรรจากค่าย joker123 เว็บไซต์หลักนั้น มาพร้อมระบบสุ่มรางวัลที่เที่ยงธรรม ไม่มีการปรับปรุงอัตราแตกแต่อย่างใด ผู้เล่นสามารถเชื่อมั่นได้ว่าได้โอกาสชนะอย่างแท้จริง เมื่อคุณเล่นที่ joker123 จะได้สัมผัสกับเกมสล็อตที่แตกง่าย แตกหนัก ไม่ว่าจะเป็นเกมยอดนิยมที่ผู้คนจำนวนมากรู้จัก หรือเกมใหม่ปัจจุบันที่เพิ่งจะเปิดตัว ข้อดีก็คือ RTP สูงโดยเฉลี่ยมักอยู่ในช่วง 95-98% ซึ่งถือว่าสูงมากในอุตสาหกรรมนี้ ยิ่ง RTP สูงเท่าไร โอกาสที่ผู้เล่นจะได้เงินรางวัลก็ยิ่งมากขึ้นเรื่อยๆแค่นั้น ทำให้การเล่นไม่เฉพาะแต่เพลิดเพลินเจริญใจ ถ้าหากแต่ยังสร้างความเชื่อมั่นว่าคุณจะได้รับผลตอบแทนที่คุ้มค่าจากการลงทุน
เมื่อคุณเล่นที่ joker123 จะได้สัมผัสกับเกมสล็อตที่แตกง่าย แตกหนัก ไม่ว่าจะเป็นเกมยอดนิยมที่ผู้คนจำนวนมากรู้จัก หรือเกมใหม่ปัจจุบันที่เพิ่งจะเปิดตัว ข้อดีก็คือ RTP สูงโดยเฉลี่ยมักอยู่ในช่วง 95-98% ซึ่งถือว่าสูงมากในอุตสาหกรรมนี้ ยิ่ง RTP สูงเท่าไร โอกาสที่ผู้เล่นจะได้เงินรางวัลก็ยิ่งมากขึ้นเรื่อยๆแค่นั้น ทำให้การเล่นไม่เฉพาะแต่เพลิดเพลินเจริญใจ ถ้าหากแต่ยังสร้างความเชื่อมั่นว่าคุณจะได้รับผลตอบแทนที่คุ้มค่าจากการลงทุน

 คุณลักษณะเด่นของ joker123 ที่คุณไม่ควรพลาด
คุณลักษณะเด่นของ joker123 ที่คุณไม่ควรพลาด

 สล็อต168 เว็บไซต์ตรง รวมเกมแตกง่าย โบนัสจัดเต็ม จังหวะชนะสูง ผลกำไรเน้นๆ
สล็อต168 เว็บไซต์ตรง รวมเกมแตกง่าย โบนัสจัดเต็ม จังหวะชนะสูง ผลกำไรเน้นๆ สล็อต168 ใหม่ล่าสุด รวมเกมยอดนิยม โบนัสแตกง่ายยิ่งกว่าเดิม
สล็อต168 ใหม่ล่าสุด รวมเกมยอดนิยม โบนัสแตกง่ายยิ่งกว่าเดิม
 มัดรวมมาให้แบบครบจบในที่เดียว! Alot66 พาคุณมาดูไม่เหมือนกันของระบบ RNG แล้วก็ค่า RTP แบบชัดๆ
มัดรวมมาให้แบบครบจบในที่เดียว! Alot66 พาคุณมาดูไม่เหมือนกันของระบบ RNG แล้วก็ค่า RTP แบบชัดๆ • RTP ไม่ได้ทำให้ชนะทันที: ค่า RTP เป็นการคำนวณผลในระยะยาว มิได้แปลว่า ผู้เล่นจะได้เงินคืนตามอัตรานี้เมื่อใดก็ตามเล่นในระยะสั้น อาจมีตอนที่ชนะและก็แพ้มากมายหรือน้อยกว่าที่คาดไว้ได้
• RTP ไม่ได้ทำให้ชนะทันที: ค่า RTP เป็นการคำนวณผลในระยะยาว มิได้แปลว่า ผู้เล่นจะได้เงินคืนตามอัตรานี้เมื่อใดก็ตามเล่นในระยะสั้น อาจมีตอนที่ชนะและก็แพ้มากมายหรือน้อยกว่าที่คาดไว้ได้

 มองหาเว็บตรงไม่ผ่านเอเย่นต์ มองหา
มองหาเว็บตรงไม่ผ่านเอเย่นต์ มองหา  • สร้างโอกาสสำหรับการทำเงินต่อ: ผู้คนจำนวนไม่ใช้น้อยบางทีอาจจะเลือกนำยอดเสียที่ได้รับคืนมาต่อยอดสำหรับในการเล่นสล็อตออนไลน์ทันที
• สร้างโอกาสสำหรับการทำเงินต่อ: ผู้คนจำนวนไม่ใช้น้อยบางทีอาจจะเลือกนำยอดเสียที่ได้รับคืนมาต่อยอดสำหรับในการเล่นสล็อตออนไลน์ทันที
 เลือกเกมสล็อตออนไลน์กับ ฟูลสล็อตพีจี ได้แบบง่ายๆให้เหมาะกับคุณด้วย 3 ขั้นตอนง่ายๆดังนี้!
เลือกเกมสล็อตออนไลน์กับ ฟูลสล็อตพีจี ได้แบบง่ายๆให้เหมาะกับคุณด้วย 3 ขั้นตอนง่ายๆดังนี้! อะไรจะดีไปกว่าการเล่นเกมที่พวกเราชอบอีกล่ะครับผมจริงไหม? เรา pg slot มีเกมสล็อตออนไลน์จากค่าย PG ให้คุณได้เลือกจำนวนมากจำนวนมาก แต่ละเกมก็จะมีธีมและก็เรื่องราวของเกมเป็นของตนเอง ทำให้ท่านสามารถเลือกเกมต่างๆได้อย่างอิสระ คนไม่ใช่น้อยที่ผมรู้จักก็มักจะเลือกเกมจากธีมเกมpgslotที่ถูกใจก่อนนี่แหละครับผม ส่วนเรื่องของเนื้อหาด้านในเกมก็ค่อยมาว่ากันอีกที เลือกจากความถูกหัวใจก่อนก็พอนะครับ ฮ่า… ซึ่งถ้าคุณเลอืกเกมPgslotจากธีมเกมที่คุณถูกใจ มันก็มีโอกาสที่จะทำให้คุณเล่นเกมนั้นๆได้นานขึ้น เพลิดเพลินใจรวมทั้งบันเทิงใจกับการเล่นสล็อตออนไลน์เพิ่มขึ้นเรื่อยๆ ซึ่งเกมจากค่าย PG ของพวกเรามีเกมสล็อตเยอะแยะและหลากหลายธีมให้ท่านเลือกแน่นอนขอรับ ถูกใจแบบไหนก็เลือกกันได้เลยค่ะขอรับ
อะไรจะดีไปกว่าการเล่นเกมที่พวกเราชอบอีกล่ะครับผมจริงไหม? เรา pg slot มีเกมสล็อตออนไลน์จากค่าย PG ให้คุณได้เลือกจำนวนมากจำนวนมาก แต่ละเกมก็จะมีธีมและก็เรื่องราวของเกมเป็นของตนเอง ทำให้ท่านสามารถเลือกเกมต่างๆได้อย่างอิสระ คนไม่ใช่น้อยที่ผมรู้จักก็มักจะเลือกเกมจากธีมเกมpgslotที่ถูกใจก่อนนี่แหละครับผม ส่วนเรื่องของเนื้อหาด้านในเกมก็ค่อยมาว่ากันอีกที เลือกจากความถูกหัวใจก่อนก็พอนะครับ ฮ่า… ซึ่งถ้าคุณเลอืกเกมPgslotจากธีมเกมที่คุณถูกใจ มันก็มีโอกาสที่จะทำให้คุณเล่นเกมนั้นๆได้นานขึ้น เพลิดเพลินใจรวมทั้งบันเทิงใจกับการเล่นสล็อตออนไลน์เพิ่มขึ้นเรื่อยๆ ซึ่งเกมจากค่าย PG ของพวกเรามีเกมสล็อตเยอะแยะและหลากหลายธีมให้ท่านเลือกแน่นอนขอรับ ถูกใจแบบไหนก็เลือกกันได้เลยค่ะขอรับ

 สล็อตเว็บตรงแท้ที่ดีเยี่ยมที่สุดในไทย เว็บตรงไม่ผ่านเอเย่นต์จาก pg slot แหล่งสรุปความสนุกสนานแบบครบถ้วน
สล็อตเว็บตรงแท้ที่ดีเยี่ยมที่สุดในไทย เว็บตรงไม่ผ่านเอเย่นต์จาก pg slot แหล่งสรุปความสนุกสนานแบบครบถ้วน • การทดลองเล่น: และก็สุดท้าย มืออาชีพจำนวนมากจะเลือกเกมอะไรสักอย่าง พวกเขาจะทดสอบเล่นเกมสล็อต456นั้นๆจนมีความรู้ความสามารถแล้วก็ความเชี่ยวชาญสำหรับเพื่อการเล่นมากพอ ซึ่งมันจะช่วยให้คุณเล่นเกมนั้นๆเจริญขึ้นอีกด้วยครับ
• การทดลองเล่น: และก็สุดท้าย มืออาชีพจำนวนมากจะเลือกเกมอะไรสักอย่าง พวกเขาจะทดสอบเล่นเกมสล็อต456นั้นๆจนมีความรู้ความสามารถแล้วก็ความเชี่ยวชาญสำหรับเพื่อการเล่นมากพอ ซึ่งมันจะช่วยให้คุณเล่นเกมนั้นๆเจริญขึ้นอีกด้วยครับ

 สล็อต888เว็บไซต์ตรง ลงทุนขั้นต่ำแค่ 1 บาท ลุ้นรับแจ็คพ็อตได้ แจ็คพ็อตแตกทุกวัน ! ไม่ล็อคยูส ไม่ล็อคผล
สล็อต888เว็บไซต์ตรง ลงทุนขั้นต่ำแค่ 1 บาท ลุ้นรับแจ็คพ็อตได้ แจ็คพ็อตแตกทุกวัน ! ไม่ล็อคยูส ไม่ล็อคผล สวัสดีครับผมทุกคน! สำหรับผู้ที่กำลังมองหาเว็บสล็อตที่ให้ท่านสนุกสนานและได้โอกาสได้รับแจ็คพ็อตแตกทุกๆวัน ผมขอชี้แนะให้คุณรู้จักกับ เว็บไซต์สล็อต สล็อต เว็บสล็อตที่มาแรงและได้รับความนิยมจากผู้เล่นเยอะแยะในขณะนี้ สิ่งที่ทำให้เว็บแห่งนี้โดดเด่นเป็นการที่คุณสามารถเริ่มต้นลงทุนขั้นต่ำเพียงแค่ 1 บาทเท่านั้นนะครับ! นอกจากการลงทุนที่น้อยนิดแต่ได้โอกาสได้รับผลตอบแทนสูงแล้ว เว็บไซต์ สล็อต ยังมากับระบบการเล่นที่เป็นธรรม ไม่มีการล็อคยูสเซอร์หรือล็อคผล ทำให้ท่านสามารถสนุกสนานกับการเล่นได้อย่างเต็มที่ โดยไม่จำเป็นต้องกลุ้มใจเรื่องการปั่นผลหรือล็อคแจ็คพ็อต
สวัสดีครับผมทุกคน! สำหรับผู้ที่กำลังมองหาเว็บสล็อตที่ให้ท่านสนุกสนานและได้โอกาสได้รับแจ็คพ็อตแตกทุกๆวัน ผมขอชี้แนะให้คุณรู้จักกับ เว็บไซต์สล็อต สล็อต เว็บสล็อตที่มาแรงและได้รับความนิยมจากผู้เล่นเยอะแยะในขณะนี้ สิ่งที่ทำให้เว็บแห่งนี้โดดเด่นเป็นการที่คุณสามารถเริ่มต้นลงทุนขั้นต่ำเพียงแค่ 1 บาทเท่านั้นนะครับ! นอกจากการลงทุนที่น้อยนิดแต่ได้โอกาสได้รับผลตอบแทนสูงแล้ว เว็บไซต์ สล็อต ยังมากับระบบการเล่นที่เป็นธรรม ไม่มีการล็อคยูสเซอร์หรือล็อคผล ทำให้ท่านสามารถสนุกสนานกับการเล่นได้อย่างเต็มที่ โดยไม่จำเป็นต้องกลุ้มใจเรื่องการปั่นผลหรือล็อคแจ็คพ็อต ในบทความนี้เราจะมาเอ๋ยถึงเหตุผลว่าเพราะอะไรการเล่นสล็อตเว็บไซต์ตรง สล็อต นั้นเป็นตัวเลือกที่ดีสำหรับผู้เล่นทั้งยังมือใหม่แล้วก็มืออาชีพ อีกทั้งยังมีวิธีการเล็กๆน้อยๆที่จะสามารถช่วยให้ท่านมีโอกาสชนะเยอะขึ้นเรื่อยๆ และไม่ลืมที่จะพรีเซ็นท์โปรโมชั่นสุดพิเศษที่คุณไม่สมควรพลาด ด้วยเหตุนั้นหากคุณเป็นผู้ที่ติดอกติดใจการเสี่ยงโชครวมทั้งอยากได้ความท้าทายที่มีความสนุกสนาน มาอ่านเนื้อหานี้จนจบ แล้วคุณจะทราบดีว่าเพราะเหตุไร สล็อต ถึงเป็นเว็บไซต์สล็อตที่คุณควรลองเล่น!
ในบทความนี้เราจะมาเอ๋ยถึงเหตุผลว่าเพราะอะไรการเล่นสล็อตเว็บไซต์ตรง สล็อต นั้นเป็นตัวเลือกที่ดีสำหรับผู้เล่นทั้งยังมือใหม่แล้วก็มืออาชีพ อีกทั้งยังมีวิธีการเล็กๆน้อยๆที่จะสามารถช่วยให้ท่านมีโอกาสชนะเยอะขึ้นเรื่อยๆ และไม่ลืมที่จะพรีเซ็นท์โปรโมชั่นสุดพิเศษที่คุณไม่สมควรพลาด ด้วยเหตุนั้นหากคุณเป็นผู้ที่ติดอกติดใจการเสี่ยงโชครวมทั้งอยากได้ความท้าทายที่มีความสนุกสนาน มาอ่านเนื้อหานี้จนจบ แล้วคุณจะทราบดีว่าเพราะเหตุไร สล็อต ถึงเป็นเว็บไซต์สล็อตที่คุณควรลองเล่น!
 3. สร้างสมดุลในการเล่นสล็อตออนไลน์ pgslot ระหว่างความสนุกสนานรวมทั้งความเสี่ยง
3. สร้างสมดุลในการเล่นสล็อตออนไลน์ pgslot ระหว่างความสนุกสนานรวมทั้งความเสี่ยง


 Pussy888 เกมสล็อตออนไลน์ ดาวน์โหลด สล็อต888 เทคนิคการเล่นสล็อตออนไลน์
Pussy888 เกมสล็อตออนไลน์ ดาวน์โหลด สล็อต888 เทคนิคการเล่นสล็อตออนไลน์ เกม pussy888 ได้เปลี่ยนเป็นหนึ่งในกิจกรรมที่ได้รับความนิยมที่ผู้คนทั้งโลกเผลอไผล และในประเทศไทยเองก็ไม่มีความแตกต่างกัน เกม pussy888 ที่ได้รับความนิยมอย่างยิ่งในปัจจุบันเป็น สล็อตพุซซี่ ซึ่งไม่เฉพาะแต่มีกราฟิกที่สวยสดงดงามและก็ฟีเจอร์ที่น่าดึงดูด แต่ว่ายังมีแนวทางการเล่นสล็อตออนไลน์ที่จะสามารถช่วยให้คุณเพิ่มช่องทางสำหรับการชนะเยอะขึ้นเรื่อยๆ
เกม pussy888 ได้เปลี่ยนเป็นหนึ่งในกิจกรรมที่ได้รับความนิยมที่ผู้คนทั้งโลกเผลอไผล และในประเทศไทยเองก็ไม่มีความแตกต่างกัน เกม pussy888 ที่ได้รับความนิยมอย่างยิ่งในปัจจุบันเป็น สล็อตพุซซี่ ซึ่งไม่เฉพาะแต่มีกราฟิกที่สวยสดงดงามและก็ฟีเจอร์ที่น่าดึงดูด แต่ว่ายังมีแนวทางการเล่นสล็อตออนไลน์ที่จะสามารถช่วยให้คุณเพิ่มช่องทางสำหรับการชนะเยอะขึ้นเรื่อยๆ
 918kiss สล็อตเว็บไซต์ตรงที่ยอดเยี่ยมในไทย รับประกันความแตกง่ายทุกเกม 100% ลงทะเบียนเป็นสมาชิกรับโปรโมชั่นได้โดยทันที!
918kiss สล็อตเว็บไซต์ตรงที่ยอดเยี่ยมในไทย รับประกันความแตกง่ายทุกเกม 100% ลงทะเบียนเป็นสมาชิกรับโปรโมชั่นได้โดยทันที! สำรวจตัวเองก่อนเล่นสล็อตออนไลน์ ถ้าหากคุณคือสายทุนน้อย พวกเรา 918kissจะพาคุณมาดูแนวทางเอง!
สำรวจตัวเองก่อนเล่นสล็อตออนไลน์ ถ้าหากคุณคือสายทุนน้อย พวกเรา 918kissจะพาคุณมาดูแนวทางเอง! 1. รูปแบบของสายทุนน้อยสำหรับในการเล่นสล็อตออนไลน์กับ 918kiss
1. รูปแบบของสายทุนน้อยสำหรับในการเล่นสล็อตออนไลน์กับ 918kiss เลือกเล่น joker123 ที่มีคุณภาพสูง ฟีเจอร์พิเศษและโบนัสที่นานัปการ
เลือกเล่น joker123 ที่มีคุณภาพสูง ฟีเจอร์พิเศษและโบนัสที่นานัปการ สล็อตออนไลน์ joker123 ได้เปลี่ยนเป็นหนึ่งในรูปแบบความสนุกสนานยอดนิยมอย่างแพร่หลาย ด้วยความสะดวกสบายสำหรับการเข้าถึงแล้วก็ช่องทางสำหรับการชนะรางวัลใหญ่ที่มีอยู่ การเล่นสล็อตเว็บไซต์ตรงที่มีการให้เครดิตฟรีเป็นทางเลือกที่น่าสนใจสำหรับผู้เล่นที่ปรารถนาเพิ่มจังหวะสำหรับการเล่นเกมโดยไม่ต้องลงทุนมาก
สล็อตออนไลน์ joker123 ได้เปลี่ยนเป็นหนึ่งในรูปแบบความสนุกสนานยอดนิยมอย่างแพร่หลาย ด้วยความสะดวกสบายสำหรับการเข้าถึงแล้วก็ช่องทางสำหรับการชนะรางวัลใหญ่ที่มีอยู่ การเล่นสล็อตเว็บไซต์ตรงที่มีการให้เครดิตฟรีเป็นทางเลือกที่น่าสนใจสำหรับผู้เล่นที่ปรารถนาเพิ่มจังหวะสำหรับการเล่นเกมโดยไม่ต้องลงทุนมาก


 เกมที่มีกราฟิกสวยแล้วก็การเล่นที่ตื่นเต้น หรือจะเป็นระบบฝาก-ถอนที่รวดเร็วและก็ไม่มีอันตราย รวมถึงโปรโมชั่นที่มากมายรวมทั้งคุ้มค่า เนื้อหานี้จะพาคุณไปรู้จักกับ pgslot อย่างประณีต ไม่ว่าคุณจะเป็นมือใหม่ที่พึ่งเริ่มหรือมือโปรที่กำลังมองหาเว็บใหม่ เนื้อหานี้จะช่วยทำให้คุณได้รับข้อมูลที่มีคุณประโยชน์เพื่อคุณตัดสินใจเลือกใช้บริการได้อย่างมั่นอกมั่นใจขอรับ
เกมที่มีกราฟิกสวยแล้วก็การเล่นที่ตื่นเต้น หรือจะเป็นระบบฝาก-ถอนที่รวดเร็วและก็ไม่มีอันตราย รวมถึงโปรโมชั่นที่มากมายรวมทั้งคุ้มค่า เนื้อหานี้จะพาคุณไปรู้จักกับ pgslot อย่างประณีต ไม่ว่าคุณจะเป็นมือใหม่ที่พึ่งเริ่มหรือมือโปรที่กำลังมองหาเว็บใหม่ เนื้อหานี้จะช่วยทำให้คุณได้รับข้อมูลที่มีคุณประโยชน์เพื่อคุณตัดสินใจเลือกใช้บริการได้อย่างมั่นอกมั่นใจขอรับ เสนอแนะ pgslot เว็บไซต์สล็อตที่กำลังฮิตติดกระแสมาแรง บริการครบครันสูงที่สุด ระบบสุดล้ำ!
เสนอแนะ pgslot เว็บไซต์สล็อตที่กำลังฮิตติดกระแสมาแรง บริการครบครันสูงที่สุด ระบบสุดล้ำ!
 1.เป็นเว็บที่มีความปลอดภัยสูง การที่คุณจะเลือกเว็บสล็อตในการเล่นเกม 168สล็อต เว็บใดเว็บไซต์หนึ่งต้องให้ความสำคัญกับระบบรักษาความปลอดภัยเพราะไม่อย่างนั้นแล้วอาจจะส่งผลให้คุณเกิดปัญหาต่างๆตามมาแล้วก็ที่สำคัญก็คือจำเป็นจะต้องได้รับการดูแลเอาใจใส่อย่างดีจาก admin มือโปรที่มีความพร้อมเพรียงแล้วก็มีสิทธิพิเศษดีๆที่จะทำให้ท่านมั่นอกมั่นใจได้ว่าเล่นเกมที่นี่แล้วคุ้มค่ารวมทั้งได้เงินเต็มปริมาณ จึงเสนอแนะว่า 168สล็อต เป็นเว็บไซต์ที่มีความรู้และความเข้าใจพิเศษที่จะสามารถช่วยให้สมาชิกเล่นเกมได้อย่างผู้เชี่ยวชาญครึกครื้นรวมทั้งสะดวกไปกับความมั่งมีมั่นคงได้เงินจริงตลอด 24 ชั่วโมงแล้วก็คุ้มค่าด้วยระบบความปลอดภัยที่ดีเยี่ยม
1.เป็นเว็บที่มีความปลอดภัยสูง การที่คุณจะเลือกเว็บสล็อตในการเล่นเกม 168สล็อต เว็บใดเว็บไซต์หนึ่งต้องให้ความสำคัญกับระบบรักษาความปลอดภัยเพราะไม่อย่างนั้นแล้วอาจจะส่งผลให้คุณเกิดปัญหาต่างๆตามมาแล้วก็ที่สำคัญก็คือจำเป็นจะต้องได้รับการดูแลเอาใจใส่อย่างดีจาก admin มือโปรที่มีความพร้อมเพรียงแล้วก็มีสิทธิพิเศษดีๆที่จะทำให้ท่านมั่นอกมั่นใจได้ว่าเล่นเกมที่นี่แล้วคุ้มค่ารวมทั้งได้เงินเต็มปริมาณ จึงเสนอแนะว่า 168สล็อต เป็นเว็บไซต์ที่มีความรู้และความเข้าใจพิเศษที่จะสามารถช่วยให้สมาชิกเล่นเกมได้อย่างผู้เชี่ยวชาญครึกครื้นรวมทั้งสะดวกไปกับความมั่งมีมั่นคงได้เงินจริงตลอด 24 ชั่วโมงแล้วก็คุ้มค่าด้วยระบบความปลอดภัยที่ดีเยี่ยม 5.มีเกมให้เลือกเยอะแยะ การเข้ามาที่ 168slot คุณไม่จำเป็นที่จะต้องมาวิตกกังวลหัวใจว่าจะมีแม้กระนั้นเกมสล็อตอย่างเดียวเพียงแค่นั้นด้วยเหตุว่าพวกเรามีเกมสนุกสนานๆให้คุณเลือกดังเช่นว่าเกมบาคาร่า เกมรูเล็ต เกม blackjack เกมป๊อกกระเด้ง เกมยิงปลา ฯลฯ ซึ่งแต่ละเกมทำให้สมาชิกสนุก รวมทั้งสร้างความเพลิดเพลินให้กับคุณได้จริงเป็นเทคโนโลยีรวมทั้งหลักการที่การบรรลุผลที่จะทำให้คุณว้าวไปกับลักษณะของเกมบันเทิงใจๆที่คุณสามารถเลือกได้ด้วยตนเอง ว่างตอนไหนก็เข้ามาเล่นเกมกับพวกเราได้เลยบันเทิงใจอย่างไม่มีข้อจำกัดจะต้องตรงนี้เท่านั้น ค้ำประกันประสิทธิภาพแล้วก็ความสุจริตใจ admin พร้อมที่จะอำนวยความสะดวกให้กับคุณอย่างยอดเยี่ยมมีคุณภาพสูงจำเป็นต้องที่นี่เท่านั้น
5.มีเกมให้เลือกเยอะแยะ การเข้ามาที่ 168slot คุณไม่จำเป็นที่จะต้องมาวิตกกังวลหัวใจว่าจะมีแม้กระนั้นเกมสล็อตอย่างเดียวเพียงแค่นั้นด้วยเหตุว่าพวกเรามีเกมสนุกสนานๆให้คุณเลือกดังเช่นว่าเกมบาคาร่า เกมรูเล็ต เกม blackjack เกมป๊อกกระเด้ง เกมยิงปลา ฯลฯ ซึ่งแต่ละเกมทำให้สมาชิกสนุก รวมทั้งสร้างความเพลิดเพลินให้กับคุณได้จริงเป็นเทคโนโลยีรวมทั้งหลักการที่การบรรลุผลที่จะทำให้คุณว้าวไปกับลักษณะของเกมบันเทิงใจๆที่คุณสามารถเลือกได้ด้วยตนเอง ว่างตอนไหนก็เข้ามาเล่นเกมกับพวกเราได้เลยบันเทิงใจอย่างไม่มีข้อจำกัดจะต้องตรงนี้เท่านั้น ค้ำประกันประสิทธิภาพแล้วก็ความสุจริตใจ admin พร้อมที่จะอำนวยความสะดวกให้กับคุณอย่างยอดเยี่ยมมีคุณภาพสูงจำเป็นต้องที่นี่เท่านั้น

 เกม
เกม





 4. หนังเกาหลี / ซีรีส์ประเทศเกาหลี
4. หนังเกาหลี / ซีรีส์ประเทศเกาหลี เป็นไงบ้างนะครับกับ มาดูหนังออนไลน์ที่ ดูหนัง EP.1 ความจริงถ้าผมไม่รวมหนังกับซีรีส์ของแต่ละประเภทเข้าด้วยกันครับผม ผมบอกเลยว่า หมู่ของเว็บดูหนังอย่างพวกเราล้นแน่นอนขอรับ ฮ่า… รวมทั้งยังมีหนังหรือซีรีส์อีกเยอะมากจำนวนมากหลายประเภทเลยจ้ะนะครับ ซึ่งผมจะมาเขียนไว้ใน EP.2 รอติดตามกันครับ สำหรับวันนี้ ผมก็จำต้องขอตัวลาไปก่อน แล้วเจอกันใหม่ใน EP.2 นะครับ สวัสดีครับ
เป็นไงบ้างนะครับกับ มาดูหนังออนไลน์ที่ ดูหนัง EP.1 ความจริงถ้าผมไม่รวมหนังกับซีรีส์ของแต่ละประเภทเข้าด้วยกันครับผม ผมบอกเลยว่า หมู่ของเว็บดูหนังอย่างพวกเราล้นแน่นอนขอรับ ฮ่า… รวมทั้งยังมีหนังหรือซีรีส์อีกเยอะมากจำนวนมากหลายประเภทเลยจ้ะนะครับ ซึ่งผมจะมาเขียนไว้ใน EP.2 รอติดตามกันครับ สำหรับวันนี้ ผมก็จำต้องขอตัวลาไปก่อน แล้วเจอกันใหม่ใน EP.2 นะครับ สวัสดีครับ

 • สล็อต pg slot เว็บเอเย่นต์ ก็คือสิ่งที่ตรงข้ามกับสล็อตpgแท้ อย่างสิ้นเชิงเลยครับผม หมายถึง เว็บไซต์สล็อตออนไลน์ที่เปิดให้บริการนั้นไม่ได้การยืนยันจากค่ายเกมสล็อตออนไลน์ต่างๆนั่นเอง เป็นเพียงแต่การดึงตัวเกมจากค่ายต่างๆมาเปิดเองแค่นั้น มันก็ทำให้ความปลอดภัยและความน่าไว้ใจต่ำนั่น้องครับผม คนใดกันแน่ที่จะเล่นสล็อตเว็บเอเย่นต์ก็ระวังกันหน่อยครับผม
• สล็อต pg slot เว็บเอเย่นต์ ก็คือสิ่งที่ตรงข้ามกับสล็อตpgแท้ อย่างสิ้นเชิงเลยครับผม หมายถึง เว็บไซต์สล็อตออนไลน์ที่เปิดให้บริการนั้นไม่ได้การยืนยันจากค่ายเกมสล็อตออนไลน์ต่างๆนั่นเอง เป็นเพียงแต่การดึงตัวเกมจากค่ายต่างๆมาเปิดเองแค่นั้น มันก็ทำให้ความปลอดภัยและความน่าไว้ใจต่ำนั่น้องครับผม คนใดกันแน่ที่จะเล่นสล็อตเว็บเอเย่นต์ก็ระวังกันหน่อยครับผม ขอขอบคุณมากweb
ขอขอบคุณมากweb 



 แทงหวยสด มือใหม่ก็แทงได้ร่ำรวยจริง game.no1huay
แทงหวยสด มือใหม่ก็แทงได้ร่ำรวยจริง game.no1huay  เพราะเหตุใดถึงควรจะมีสลากกินแบ่งออนไลน์ แทงหวยสด
เพราะเหตุใดถึงควรจะมีสลากกินแบ่งออนไลน์ แทงหวยสด

 pg slot
pg slot  ทางเข้า pg เกมสล็อตเล่นง่าย pg slot
ทางเข้า pg เกมสล็อตเล่นง่าย pg slot